Journal of
Threatened Taxa | www.threatenedtaxa.org | 26 December 2018 | 10(15):
12907–12915
Dietary preference and feeding patterns of the urban Rhesus Macaque Macaca mulatta (Mammalia: Primates: Cercopithecidae) in Asola-Bhatti
Wildlife Sanctuary in India
Ishita Ganguly 1 & Netrapal Singh Chauhan 2
1,2 Amity Institute of Forestry and Wildlife,
Amity University Campus, Sector 125, Gautam Buddha
Nagar, Noida, Uttar Pradesh 201303, India
1 ishitaganguly23@gmail.com (corresponding
author), 2 nschauhan@amity.edu
Abstract: We studied the feeding patterns and discrete
spatio-temporal food habitsof16 groups of the urban
Rhesus Macaque Macaca mulatta
following their relocation in Asola-Bhatti Wildlife
Sanctuary near Delhi, India. We observed
that the macaques fed on 31 plant species, with Prosopis
juliflora and P. cineraria appearing in
most scans. We classified the food
consumed by the species into six main categories the species and recorded the
average time spent on each of these throughout the year. The maximum time was spent on supplementary
feeding provided by the forest department and the minimum on natural plant
resources. There was a significant
difference in the consumption of different food categories from morning to
evening but there were no significant seasonal variations. This study showed that Rhesus Macaque adopted
different foraging strategies based on the availability of resources in their
new environment and that variety in food resources buffered seasonality in
their diet. Information on their feeding
patterns and food habits will help in developing management protocols for the
primates in urban environments.
Keywords: Feeding ecology, Delhi NCR, management,
opportunistic feeding, primates, relocation, urban landscape.
doi: https://doi.org/10.11609/jott.4347.10.15.12907-12915
Editor: Mewa Singh, University of Mysore, Mysuru, India Date of publication: 26 December 2018 (online & print)
Manuscript details: Ms
# 4347 | Received 20 June 2018 | Final received 04 December 2018 | Finally
accepted 19 December 2018
Citation: Ganguly, I
& N.S. Chauhan (2018). Dietary preference and feeding patterns of
the urban Rhesus Macaque Macaca mulatta (Mammalia: Primates: Cercopithecidae)
in Asola-Bhatti Wildlife Sanctuary in India. Journal
of Threatened Taxa 10(15): 12907–12915; https://doi.org/10.11609/jott.4347.10.15.12907-12915
Copyright: © Ganguly & Chauhan 2018. Creative Commons
Attribution 4.0 International License. JoTT
allows unrestricted use of this article in any medium, reproduction and
distribution by providing adequate credit to the authors and the source of
publication.
Funding: This study was funded by World Wide Fund
for Nature - Small Grant Programme (2016–2018), India.
Competing interests: The authors declare no competing interests.
Author
Details: Ishita Ganguly, B.Sc (Hons.), MSc, MPhil in Zoology and presently
pursuing doctoral degree in Wildlife Science from Amity Institute of Forestry
and Wildlife, Amity University Noida, India. I have been working on ecology of
Rhesus Macaque and human-macaque conflict in urban landscape since 2015. My
project is funded by WWF Small grant programme
(2016–2018). Currently, writing my thesis and also preparing
for attending students conference (SSCS Cambridge, UK) 2019 in United Kingdom. Dr.
N.P.S. Chauhan, MSc & PhD, Zoology from
Delhi University, actively involved in teaching, training and research in Delhi
University colleges, North-Eastern Hill University, Shillong,
Wildlife Institute of India, Dehradun and now in Amity University, Noida and
presently serving as Director of Amity Institute of Forestry and Wildlife. He
is the main supervisor of many PhD students in Amity University.
Author Contribution: IG wrote the project, raised funding,
completed field research, worked on data analysis, writing manuscript and
communication. NSC contributed in planning of research, writing the manuscript
and revising.
Acknowledgements: We thank Mr. A.K. Shukla,
Chief Wildlife Warden,Delhi
Forest and Wildlife Department, for issuing the permit to work in the field
area and Mr. S.K. Muan Giete, Deputy Conservator of Forest, South Division, Delhi,
for providing support for the smooth conduct of the field research. We also
thank Prof. Dr. Praful Singh and Ms. Pradipika Verma, Amity Institute
of GIS and Remote Sensing,for
designing the maps though GIS software and data imaging and Dr.
G.S. Rawat, Wildlife Institute of India, for
identifying the plant species specimens.
Introduction
Rhesus
Macaque Macaca mulatta
is the most common non-human primate in the forested and urban areas of Asia (Hasan et al. 2013).
It is found throughout India in its peninsular (Madhya Pradesh, West
Bengal, and Assam), northern (Jammu & Kashmir, Himachal Pradesh, Punjab,
Haryana, Uttar Pradesh, Rajasthan, and Gujarat) (Seth et al. 2001), and northeastern (Assam, Meghalaya, and Arunachal Pradesh; (Molur et al. 2003) regions.
Information on the feeding ecology of a species provides the detailed
dietary specialization necessary for its survival and is an important part of
its natural life history (Harcourt et al. 2002). Flexibility in diet patterns
plays a pivotal role in the survival of non-human primate species in urban and peri-urban ecosystems through resource sharing and
competition and has evolutionary implications in the long-term. Primates are
known to adopt several foraging strategies (Fleagle
& Gilbert 2006). Natural diet of forest Rhesus Macaques includes fruits,
seeds, inflorescences, flowers, buds, leaves, young shoots, twigs, barks,
roots, and pith and resin of gymnosperms, angiosperms, and fungi (Fooden 2000). Macaques are also known to consume animal
food items that such as insects, spiders, worms, termites, grasshoppers,
lizards, ants, beetles, molluscs, crayfish, shellfish, honeycombs, crabs, and
bird eggs (Mandal 1964; Lindburg
1971; Malik 1983). In marine coastal areas, the species is mostly known to rely
on seeds and fruits (Hanya et al. 2003) and also
catch live fish as in the Sunderban (Majumder et al. 2012). In forested habitats,primates consume 25% to 40% of the total frugivore biomass (Chapman et al. 1995). Ingestion of
fruits or young leaves with sugar and insects with protein content help to
balance their diet (Janson & Chapman 1999).
Feeding patterns are also associated with human-macaque conflict—crop raiding
by macaques in villages near forest areas has increased the level of negative
association of the species by farmers (Air 2015). The dependency of urban macaques on anthropogenic
food resources and their behaviours associated with food utilisation from urban
areas often increase the risk of undesirable interactions with human beings (Sha & Hanya 2013).
In
urban landscapes, the nutrition required for primates becomes highly
questionable. Urban macaques largely share human food resources (Gupta 2001)
and depend on cultivated crops, plants, and even garbage (Lee et al. 1986). In
some cases, macaques depend on humans for being fed (Strum 1994)—the feeding
patterns of primates that live in tourist sites and temples are often influenced
by provisioning of food by humans. Urban macaques have also acquired
behavioural adaptations in food-acquisition techniques (Mangalam
& Singh 2013). Urban habitats, in contrast to natural ones, have a more
direct influence on primate behaviours associated with competitive resource
utilizations and foraging techniques. Several anthropogenic barriers and
disturbances interfere with the feeding ecology of primates in urban
environments.
Although
Rhesus Macaques were assessed as a Least Concern (LC) species by IUCN (2018),
primates are threatened globally by human-wildlife negative interactions,
habitat loss and fragmentation, and several other anthropogenic factors (Strum
1994, 2001; Mittermeier & Konstant
1996, 1997; Kemf & Wilson 1997; Cowlishaw & Dunbar 2000; Peterson 2003; Hill
2005). The Negative interactions between
humans and macaques due to food provisioning and other anthropogenic drivers
possess major challenges for the survival and persistence of the species. The
need for translocation of Rhesus Macaques and the consequences were due to its
proliferation in urban areas of India was suggested and studied earlier (Malik
& Johnson 1991, 1994; Southwick et al. 1998). Translocation or relocation
is a widely used conservation tool but it is known to induce stress, as evident
in the higher level of stress hormones in females of the species during the
translocation process (Aguilar-Cucurachi et al.
2010).
The
reproductive capacity, inter-birth interval, and the size of social groups in
primates are often determined by the amount of food they consume (Air
2015). Again, the availability of
different food resources can reduce seasonal fluctuations in diet and
provisioning of food regularly to urban primates may have adverse effects on
their behaviour, social organization, and conservation (Sinha&Vijayakrishnan
2017). The aim of this study was to investigate the food habits and feeding
patterns of urban macaques in and around (0–1 km) Asola-Bhatti
Wildlife Sanctuary in Delhi. The characteristic features of this sanctuary such
as the availability of food resources infringe villages and food provisioning
by the public have played an influential roles in the feeding ecology of its
Rhesus Macaques. Information of dietary patterns of urban macaques will enhance
the knowledge of its natural history and survival and that will help in the
management of the species in urban ecosystems.
Materials and
methods
Study
area
Asola-Bhatti Wildlife Sanctuary is situated in
South Delhi District (28.410–28.490 N and 77.190N–77.270
E) and covers a total area of about 6,874ha and there is a high density of Acacia pendula,scrub
forests, and trees with a short diameters (Kushwaha
et al. 2014) (Fig. 1). The forest
area is located at the foothills of the Aravalli
range and is about 16km long and 4.3km wide, with elevation ranging from
235–288 m. The forest is surrounded by hilly areas with shrubs, stunted trees,
and moderate density forest cover and is adjacent to the urban areas of
Delhi-Haryana interstate border region, Sangam Vihar, Faridabad (Surajkund
Road), Pali Village, Satberi,
Deragaon, Fatepurberi, and Anangpur. This
protected area has semi-arid vegetation with xerophytic
plants and several large, deep pits. The
largest water-filled pit is Neeli-Jheel, situated 3km
from Gate No. 7, where urban monkeys are intermittently released after capture
since 2007.
The dominant tree species in Asola-Bhatti
Wildlife Sanctuary are Prosopis juliflora, a native species introduced to counter
forest degradation (Burkart 1976; Pasiecznik
et al. 2001), which and present at high (7.68%), moderate (16.03%), and
low (47.90%) densities,and Anogeissus pendula
and Acacia nilotica, present in forest
plantation, scrub vegetation (12.04%), water bodies (0.16%), and human
settlements (2.92%) (Kushwaha et
al. 2014). There were no Rhesus
Macaques in the area before translocation began and the present population is
derived entirely from relocated animals alone.
This area exhibits extreme fluctuation in annual temperature, with
summer highs in May (43–47 0C)
and winter lows in January (60C). June to September is the wet season with an average annual rainfall
of about 617mm. This sanctuary is
composed of Asola Village in the north and Bhatti area in the south. It is a man-made sanctuary and
the only protected area in Delhi. Most
of the area of the sanctuary is degraded with the prevalence of xerophytic plants (Khanna &
Sati 2003). Vegetation shows remarkable dominance of shrubs and stunted trees (Naithani et al. 2006).
Study groups
We
sighted a total of 16 groups of translocated Rhesus
Macaques in the area. Observations were made from a close distance of <10m
and data on demography and food habits were collected from 06.00–18.00 hr daily from May 2016 to June 2017. Group size varied from 14 to 63 individuals
per group (30.57±2.67) and a total of 492 (n) individuals were observed (Table
1).
Food categories
We classified the food consumed by the relocated Rhesus Macaques into
categories based on variations in resource availability:
Natural plant species: The natural plants, trees, herbs, and shrubs
available in the sanctuary.
Supplementary foods: Food given by the forest department daily in this
sanctuary for feeding the Rhesus Macaques only (seasonal vegetables and fruits;
2500kg/day).
Provisioned food: Banana and
roadside food (bread and chick-pea) thrown by the public to the macaques daily;
the macaques often travel to the boundary walls, cross it, and sit on the
highway it to have these itemsthe the.
Anthropogenic food: Garbage and human food resources (Indian bread, oily
fries, and potato chips) snatched by the macaques daily from human settlements
situated within 0.5km of the protected area.
Water: Water from channelsmade in the
sanctuary exclusively for Rhesus Macaques (a total of 36 in number).
Others: Insects, soil, lizards, and bird eggs.
Scan sampling
technique
Instantaneous
or scan sampling (Altmann 1974) was used to gather
information on the feeding habits and food items of the macaques. Group scans were taken on all visible members
of the group for 5min at every 10-minute interval. We recorded 13,740 scan samples
and the type of food items eaten (young leaves,mature leaves, roots, stems, flowers, fruits,
shoots, gum, bark, or animal prey). We
collected the unidentified species for taxonomic identification (leaves, stem,
and fruits) through herbarium in the Wildlife Institute of India, Dehradun.
Focal sampling technique
We
focused on individuals (adult male/ adult female/ juvenile/ infant) and made 12
entries per hour of theiractivitiesins. We recorded the time spent by that focal individualson each food plant and the parts eaten along
with thetime spent at different feeding sites. We
recorded 13,874 focal samples and categorized the different plant parts eaten
by the macaques.
We
estimated the time spent feeding on different food items in a day as per theformula by Gupta &Kumar (1994):
Ta
= Na / N x 100,
where Ta
is the percentage of time spent on an activity a, Na is the number
of records with activity a, andN is the total number
of records for the day.
Analysis:
Analysis of variance (ANOVA) was used to compare the feeding time on food
categories and the number of food plants eaten monthly and seasonally (Simpson
et al. 1960). Independent sample t-test
was used to analyse the difference in the average time spent on each
category. Chi square test was performed
to compare the association between groups.
Microsoft Excel 2010 was used to summarize the data and Minitab version
17.0 software and web tool were used to calculate descriptive statistics.
Landsat data imageries 2016 and ArcGIS software were used to map the study area
using coordinates collected during the data sampling though Garmin GPS 72H.
Results
Food categories
Food
plants, plant families, parts eaten, and the average percentage of time spent
feeding on each plant species are given in Table 2. The macaques were mostly found to feed on Fabaceae (8.76±2.64), Moraceae
(2.60±2.06), Rhamnaceae (0.34±0.02), and Mytraceae (0.06±0.03) families. Among the plant parts
(nature food items) eaten, 34.65% of feeding time was spent on leaves, followed
by 31% on bark and piths, 22.90% on flowers, and 11.01% on fruits. The macaques were mostly found in the lower
canopy and bottom of trees in summer (39.13%), in the upper canopy in monsoon
(31.26%), and in the middle to lower canopy in the winter (19%). The species was observed to spend 79% time on
the ground and only 21% time on the trees.
We
investigated the spatio-temporal feeding pattern of
Rhesus Macaques in the sanctuary (Fig. 2). Daily percentage time spent on
consuming different food categories (mentioned above) varied significantly with
time intervals from 06:00–18:00 hr. Macaques adopted their feeding strategy to
access all kind of resources available but with distinct time management
practice. On average natural plant
species eaten was calculated (mean±SE) 22.13±6.60,
provisioned food 14.63±3.53, supplementary food by forest department 35.2±10.2,
anthropogenic food resources 37.88±1.49, water 9.46±1.13 and others (insects,
birds’ eggs, lizards etc.) 6.02±0.60 and one-way ANOVA analysis showed significant
difference in percent time spent on various food
categories per day (F=4.09, df=5, P=0.01). The maximum time was spent on bananas
(31%), followed by seasonal vegetables (27%), fruits (13.07%), bread (8.02%),
garbage (7.8%), and icecreams (6%) and differed
significantly (t=3.63, df=5, P=0.01).
Seasonality and Diet
Average
percentage time spent on each food category was calculated for each month. The
overall mean time spent on natural plant species was 13.29±2.32, in
supplementary feeding provided by forest department 50.19±3.49, in
anthropogenic food category 18.18±1.41, and in provisional food by public
18.34±4 throughout the year including summer, monsoon, and winter months (Table
3). We recorded the dietary pattern and
found that the maximum average time was spent on supplementary food in all
seasons followed by a maximum on provisioned food in winter (26%), in summer
(18%), and a minimum during monsoon (11%). The macaques were observed spending
maximum time consuming natural plant species (17.68%) during the rainy season,
apart from supplementary food. ANOVA
analysis showed that there was no statistically significant difference in the
total dietary intake pattern throughout the year (F=0.05, df=11,
P>0.05).
Age-sex feeding pattern in groups
We
recorded the percentage of time spent by individuals in a group on each food
type (Table 4). We calculated the
average percentage time spent on all food categories by adult males
(25.0±8.17), adult females (22.01±7.13), sub-adult males (7.92±4.02), sub adult
females (6.06±3.19), juveniles (0.65±0.20,) and infants (0). Adult males dominated the pattern and used up
the maximum amount of food provided to them and spent the maximum time on it.
Adult females were much protective and did not allow their infants to feed on
artificial foods. Infants compensated
their nutritional requirement through lactation only.
Discussion
Non-human
primates compete with human beings for resource utilisation and space, which
can lead to negative interactions (Priston & Underdown 2009), especially in urban areas (Lee & Priston 2005). In
India, Rhesus Macaques often co-exist with human populations and are highly
dependent on them for food (Southwick et al. 1976). The high feeding dependency on anthropogenic
food resources is, however, not correlated with natural resource scarcity. While
natural resources such as fruits are highly variable over the year,
anthropogenic food resources are potentially more stable and easily available.
A study on Long-tailed Macaques Macaca fascicularis showed that the main drivers for
exploitation of anthropogenic foods were natural food plant resource scarcity
or an overt dependence on anthropogenic foods (Sha
& Hanya 2013).
Utilization of anthropogenic food resources lowered preferences of
macaques on fruits and natural plants in another study (Hambali
et al. 2014). The consequences of the dependency of macaques on human food
resources can include food stealing, which may lead to negative interactions
with humans. In our study, the relocated Rhesus Macaques were more inclined
towards anthropogenic, supplementary, and provisioned food resources than
natural plant resources in the forest. As this sanctuary is situated in a
human-dominated landscape and human settlements are located close by (less than
50m away), Rhesus Macaques disperse from the sanctuary and consume food from
nearby households, markets, and temple areas. The forest department of Delhi
Government was also assigned to provide supplementary food to the rehabilitated
macaques for the maintenance of a viable population in the newly introduced
environment.
Our results showed that the macaques fed on natural plant species in the
early morning between 06:00– 09:00 hr (40.8%), after
which their tendency to consume natural resources declined before rising in the
late afternoon (21.75%). Food
provisioning by the public was recorded mostly in the early morning (25%) and
continued throughout the day in the fringes of the sanctuary. Between 09:00hr
and 11:00hr, macaques gathered at feeding stations within the sanctuary near
the Bhatti Range Office, reaching a peak number
between 12:00hr and 15:00hr (53.75%).
The macaques were reportedly given 2,500kg food per day by the forest
department and this feeding pattern had a large influence on their daily
activity and movement. During
supplementary feeding, the macaques did not consume natural plants within the
forest area. The relocated Rhesus
Macaques were highly inclined towards human food outside the sanctuary and
often entered nearby houses or snatched bread and vegetables from open markets
in the nearby Sanjay Colony (Bhatti Mines).
Our results showed that the macaques mostly preferred leaves and stems
of Prosopis juliflora
(16.34%) and P. cineraria (11.09%), which were reported to be
beneficial for their health. The
heartwood of these two plant species contains ample antioxidants such as flavonol and mesquitol (Sirmah et al. 2009).
Though numerous species of medicinal plants and fruiting trees are
available in the sanctuary, the macaques did not spend much time in natural
foraging but mostly depended on artificial feeding. Our results showed a high consumption of
supplementary food items throughout the year with no seasonal differences and
low average time spent on natural food plants.
The macaques showed dependency on anthropogenic and provisioned food
over natural fruit. The former included
bananas, seasonal fruits and vegetables, bread, chickpeas, fried snacks, and ice-creams; the macaques were even reported to steal cold
water from refrigerators of houses in nearby localities at a 0.25–5 km distance
(USA Today 2017).
Roadside
food provisioning is a common practice across cities and villages in India.
Southwick et al. (1976) documented the impact of artificial feeding on
the ecology and behaviour of macaques.
Our study provides information on feeding practices of Rhesus Macaques
after translocation to an area containing various types of natural and
anthropogenic food resources in a human-dominated landscape. An understanding of the basic natural history
of primates is essential for their conservation (Caro 2007; Fashing
2007). The primary threat primates face today is habitat destruction (Wieczkowski 2004; Chapman et al. 2006). By reducing forest
size and quality, habitat destruction leads to the reduction of food sources
for forest-dwelling primates and, in some cases, threatens them with local
extinction (Lee &Hauser 1998; Muoria et al.
2003). The increasing population of
Rhesus Macaques living in proximity to human habitations has become a major
issue in India. Rapid urbanisation,
deforestation, and habitat fragmentation altered the natural living spaces of
animals and their natural behaviour in the wild. Most primate species were severely affected
by threats in anthropogenic landscapes (Sinha & Vijayakrishnan 2017).
The translocation of Rhesus Macaques from city areas
to forest situated at close proximity with human settlementsmight
not reduce the conservation threats for the taxa. The step, however, altered their feeding
strategies as the species was observed to become more dependent on
supplementary and anthropogenic food resources than on natural foraging. Though artificial feeding of fruits and
vegetables might increase overall nutrition, their natural frugivorous
behaviour seem to be lost. The macaques were seen to snatch and steal
even those anthropogenic food resources that had no health benefits, as they
were used to such behaviour in human habitats (Ganguly
et al. 2018). The dietary habits of
Rhesus Macaques were totally different in a human-dominated forest
land. Previous studies showed that
the species thrived in eight diverse habitats (temple, urban, village,
village-cum-pond, pond, roadside, canal sides, and forest) having varying
degrees of human interactions in India (Seth et al. 1986). The feeding practice
seemed to increase the urban threats, diseases, and anthropogenic stress in the
Rhesus Macaque population. In our study, the species was observed to spend
maximum time on the ground instead of on the trees and their dependency on
supplementary, provisioned, and anthropogenic food sources did not indicate
conservation success. Understanding the feeding ecology in this sanctuary would
help in planning the management of macaques in other urban areas.
References
Aguilar-Cucurachi,
M.A., P.A.D. Dias, A. Rangel-Negrín, R. Chavira, L. Boeck & D.
Canales-Espinosa (2010). Preliminary evidence of accumulation of stress during translocation in
mantled howlers. American Journal of Primatology 72(9): 805–10; https://doi.org/10.1002/ajp.20841
Air, A. (2015). Crop Raiding and Conflict: Study of
Rhesus Macaque-human Conflict in Shivapuri-Nagarjun
National Park, Kathmandu Nepal. Natural Resources Management,
Norwegian University of Science and Technology, 32pp.
Altmann, J. (1974). Observational study
of behavior: sampling methods. Behavior
49(3/4): 227–267.
Begum, S. & M.M. Feeroz
(2013).
Distribution of Rhesus Macaques (Macaca mulatta) in Bangladesh: Interpopulation
Variation in Group Size and Composition. Primate
Conservation 26: 125–132.
Burkart, A. (1976). A monograph of the genus Prosopis (Leguminosae: Mimosoideae) (Part 1 and 2). Catalogue of the
recognized species of Prosopis. Journal of the Arnold Arboretum 57: 219–249, 450–525.
Caro, T. (2007). Behavior and
conservation: a bridge too far? Trends in Ecology &
Evolution 22(8): 394–400.
Chapman, C.A., M.J. Lawes
& H.A.C. Eeley (2006). What hope for African primate diversity? African Journal of Ecology 44: 116–133.
Chapman, C.A., R.W. Wrangham
& L.J. Chapman (1995).Ecological constraints on group-size: an analysis of
spider monkey and chimpanzee subgroups. Behavioral
Ecology and Sociobiology 36(1): 59–70.
Cowlishaw, G. &
R. I. Dunbar (2000). Primate
Conservation Biology. University of Chicago Press, Chicago, 498pp; https://doi.org/10.1017/S1367943001221337
Fashing, P.J. (2007). Behavior,
ecology and conservation of colobine monkeys: an
introduction. International Journal of Primatology 28:
507–511.
Fleagle, J.G. &
C.C. Gilbert (2006). The biogeography of primate evolution: the role of plate tectonics,
climate and chance, pp. 375–418. In: Lehman, S.M. & J.G. Fleagle (eds.). Primate Biogeography. Springer, New York.
Fooden, J. (2000). Systematic Review of the Rhesus Macaque, Macaca mulatta (Zimmermann 1780).Field Museum of Natural History, Chicago, USA,
180pp.
Ganguly, I., N.P.S. Chauhan
& P. Verma (2018).Assessment of human-macaque conflict and possible
mitigation strategies in and around Asola-Bhatti
Wildlife Sanctuary. Environment & Ecology 36(3):
823–827.
Goldstein, S.J. & A.F. Richard (1989). Ecology of Rhesus Macaques (Macaca mulatta) in
northwest Pakistan. International Journal of Primatology 10(6):
531–567.
Gupta, A.K. & A. Kumar (1994). Feeding ecology and conservation of the Phayre’s Leaf Monkey Presbytis
phayrei in northeast India. Biological
Conservation 69(3): 301–306
Gupta, A.K. (2001). Status of primates
in Tripura. Envis Bulletin 1(1): 127–135.
Hambali, K., A. Ismail, B.M. Md-Zain,
A. Amir & F.A. Karim
(2014). Diet of Long
Tailed Macaques (Macaca fascicularis)
at the Entrance of Kuala Selangor Nature Park (Anthropogenic Habitat): Food
Selection that Leads to Human-Macaque Conflict. Acta Biologica
Malaysiana 3(2): 58–68.
Hanya, G., N. Noma
& N. Agetsuma (2003). Altitudinal
and seasonal variations in the diet of Japanese Macaques in Yakushima. Primates 44: 51–59.
Harcourt, A.H., S.A. Coppeto
& S.A. Parks (2002). Rarity, specialization and extinction in primates. Journal of Biogeography 29(4): 445–456;
https://doi.org/10.1046/j.1365-2699.2002.00685.x
Hasan, K., M.A. Aziz, S.M.R. Alam, Y. Kawamoto, Y. Lisa, Jones-Engel, R.C. Kyes, S. Akhtar & C.M. Hill
(2005). People,
crops, and primates: a conflict of interests, pp. 41–59. In: Paterson,
J.D.& J. Wallis (eds.). Commensalism and Conflict: The Human–Primate
Interface. American Society of Primatologists, Norman.
IUCN (2018). The IUCN Red
List of Threatened Species, Version 2018-1. http://www.iucnredlist.org.
Downloaded on 05 July 2018.
Janson, C.H. &
C.A. Chapman (1999). Resources
and primate community structure, pp. 237–268. In: Fleagle,
J.G., C. Janson & K.E. Reed (eds.). Primate
Communities. Cambridge University Press, Cambridge.
Kemf, E. &
A. Wilson (1997). Great Apes in the Wild: Species Status Report. World Wide Fund
for Nature, Gland, 36pp.
Khanna, V. &
J.P. Sati (2003). Some Selected Fauna of Asola-Bhatti Wildlife
Sanctuary (Delhi), pp. 1–70. In: Fauna of Conservation Area Vol 16. Zoological Survey of India,
Kolkata.
Kushwaha, S.P.S., S.
Nandy & M. Gupta (2014). Growing stock & woody biomass
assessment in Asola-Bhatti Wildlife Sanctuary, Delhi,
India. Environment Monitoring Assessment 186(9): 5911–5920; https://doi.org/10.1007/s10661-014-3828-0
Lee P.C. & N.E.C. Priston
(2005). Human
attitudes to primates: Perceptions of pests, conflict and consequences for
primate conservation, pp. 1–23. In:
Paterson JD, Wallis J, editors. Commensalism and conflict: the human-primate
interface. Norman, Oklahama, American Society of
Primatology; http://doi.org/10.1002/047001539X.ch1
Lee, P.C. & M.D. Hauser (1998). Long-term
consequence of changes in territory quality on feeding and reproductive
strategies of Vervet Monkeys. Journal of Animal
Ecology 67: 347–358.
Lee, P.C., E.J. Brennan, J.G. Else &
J. Altmann (1986). Ecology and behavior
of vervet monkeys in a tourist lodge habitat, pp.
229–235. In: Else, J.G. & P.C. Lee (eds.). Primate Ecology and
Conservation. Cambridge, Cambridge University Press.
Lindburg, D.G. (1971). The Rhesus Monkey in
north Indian ecological and behavioral study, pp.
1–106. In: Rosenblum, L.A. (ed.). Primate Behavior 2. New York, Academic Press.
Majumder, J., R. Lodh
& B.K. Agarwala (2012). Fish feeding adaptation by Rhesus Macaque
Macaca mulatta
(Cercopithecidae) in the Sundarban
mangrove swamps, India. Journal of Threatened Taxa 4(4): 2539–2540;
https://doi.org/10.11609/JoTT.o2884.2539-40
Malik, I. & R.L. Johnson (1991). Trapping and conservation: development of
a translocation in India, pp. 63–64. In: Ehara, A.,
T. Kimura & M. Iwamoto (eds.). Primatology Today. Elsevier,
Amsterdam.
Malik, I. & R.L. Johnson (1994). Commensal Rhesus in India: the need and
cost of translocation. Revue d’Ecologie
(La Terre et la Vie) 49: 233–243.
Malik, I. (1983). A study of
selected behavioural traits of Rhesus Monkeys (Macaca
mulatta) in free-ranging environments. PhD
Thesis. University of Meerut.
Mandal, A.K. (1964). The
behaviour of the Rhesus Monkeys (Macaca mulatta Zimmermann) in the Sundarbans. Journal of the
Bengal Natural History Society 33: 153–165.
Mangalam, M. &
M. Singh (2013). Flexibility in food extraction techniques in urban free-ranging Bonnet
Macaques, Macaca radiata. PLoS
ONE 12: e85497;
https://doi.org/10.1371/ journal.pone.0085497
Mittermeier, R. &
W. Konstant (1996/1997). Primate conservation: a retrospective and
a look into the 21stCentury. Primate
Conservation 17: 7–17.
Molur, S., D. Brandon-Jones, W. Dittus, A. Eudey, A. Kumar, M.
Singh, M.M. Feeroz, M. Chalise,
P. Priya & S. Walker (2003). Status of South Asian Primates:
Conservation Assessment and Management Plan (C.A.M.P.) Workshop Report, 2003.
Zoo Outreach Organisation / CBSG-South Asia, Coimbatore, India, viii+432pp
Muoria, P.K., G.M. Karere,
N.N. Moinde & M.A. Suleman
(2003). Primate census and habitat evaluation in the Tana
delta region, Kenya.African Journal of
Ecology 41(2): 157–163.
Naithani, H.B., S.S. Negi,
M. Pal, S. Chandra & V.P. Khanduri (2006). Vegetation Survey and Inventorisation of Species in the Ridge Forest of Delhi.
Department of Forest and Wildlife, Government of Delhi,
Delhi.
Pasiecznik, N.M., P. Felker,
P.J.C. Harris, L.N. Harsh, G. Cruz, J.C. Tewari, K. Cadoret & L.J. Maldonado (2001). The Prosopis juliflora-Prosopis
pallida Complex. Coventry, HDRA, UK, 172pp.
Peterson, D. (2003). Eating Apes. University of
California Press, Berkeley, 320pp.
Priston, N.E.C.
& S.J. Underdown (2009). A simple method for calculating the
likelihood of crop damage by primates: an epidemiological approach. International
Journal of Pest Management 55(1): 51–56; https://doi.org/10.1080/09670870802450268
Seth, P.K. & N.K. Pokwal
(2001).Behaviour and ecology of primates. Journal of the Bombay Natural History Society 14(3): 172–180.
Seth, P.K., P.K. Chopra & S. Seth
(2001). Indian
Rhesus Macaque: habitat, ecology and activity patterns of naturally occurring
populations, pp. 68–80. In: Gupta, A.K. (ed.). Non-human
Primates of India, Vol 1(1). ENVIS
Bulletin: Wildlife & Protected Areas. Wildlife Institute
of India, Dehradun (India).
Seth, P.K. & S. Seth (1986). Ecology and behavior
of rhesus monkeys in India, pp. 89–103. In: lse, J.G.
& P.C. Lee (eds.). Primate Ecology And Conservation. Cambridge, Cambridge University Press.
Sha, J.C.M.
& G. Hanya (2013). Temporal food resource correlates to the behavior and ecology of food-enhanced Long-Tailed Macaques
(Macaca fascicularis).
Mammal Study 38(3): 163–175; https://doi.org/10.3106/041.038.0305
Simpson, G.G., A. Roe & R.C. Lewontin (1960). Quantitative Zoology. Harcourt, Brace & Company, New York.
American Journal of Science 258(2): 300–311.
Sinha, A. &
S. Vijayakrishnan (2017). Primates in Urban Settings, pp. 1–8. In:
Fuentes, A. (ed.). The International Encyclopedia
of Primatology. John Wiley & Sons, Inc. Published 2017 by John
Wiley & Sons, Inc.; https://doi.org/10.1002/9781119179313.wbprim0458
Sirmah, P., S. Dumarcay, E. Masson & P. Gerardin
(2009). Unusual amount of (-) mesquitol
from the heartwood of Propopis juliflora. Natural
Product Research 23(2): 183–189.
Southwick, C.H., I. Malik & M.F. Siddiqi (1998). Translocations of Rhesus Monkeys in
India: prospects and outcomes. American Journal of
Primatology 45(2): 209–210.
Southwick, C.H., M.F. Siddiqi,
M.Y. Farooqui & B.C. Pal (1976). The effects
of artificial feeding on aggressive behaviour of Rhesus Monkeys in India. Animal Behaviour 24(1): 11–15.
Strum, S.C. (1994). Prospects for
management of primate pests. Revue d’Ecologie (La Terre et la Vie) 49: 295–306.
Strum, S.C. (2001). Almost Human. University of Chicago
Press, Chicago, 308pp.
USA Today (2017). Why India is going bananas over birth
control for monkeys.
https://www.usatoday.com/story/news/world/2017/05/11/monkeys-india
Wieczkowski, J. (2004). Ecological correlates of abundance in the
Tana Mangabey (Cercocebus galeritus).
American Journal of Primatology 63: 125–138.
Wilson, E.O. (2007). Rhesus Monkey (Macaca mulatta)
feeding behaviour and diet of Rhesus Monkey in Shivalik forest
in north India. IUCN Red List of
Threatened Species 1(8): 1–11.
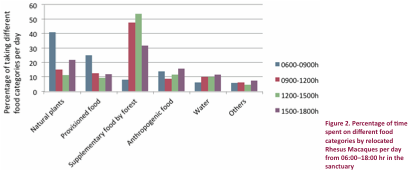
Table 1. Group composition of the
relocated Rhesus Macaques followed for studying feeding habitsin
Asola-Bhatti Wildlife Sanctuary in Delhi
|
|
AM |
AF |
SAM |
SAF |
JUV |
INF |
Total |
|
1 |
3 |
4 |
7 |
8 |
5 |
2 |
33 |
|
2 |
2 |
3 |
8 |
7 |
6 |
4 |
37 |
|
3 |
2 |
3 |
2 |
3 |
3 |
1 |
14 |
|
4 |
3 |
4 |
5 |
9 |
5 |
4 |
33 |
|
5 |
2 |
3 |
7 |
9 |
3 |
1 |
32 |
|
6 |
3 |
4 |
6 |
8 |
4 |
2 |
33 |
|
7 |
4 |
6 |
7 |
9 |
4 |
2 |
36 |
|
8 |
3 |
5 |
5 |
7 |
5 |
3 |
28 |
|
9 |
7 |
11 |
14 |
13 |
11 |
7 |
63 |
|
10 |
5 |
7 |
6 |
9 |
3 |
1 |
32 |
|
11 |
2 |
3 |
7 |
11 |
2 |
3 |
34 |
|
12 |
3 |
5 |
5 |
8 |
2 |
3 |
26 |
|
13 |
3 |
4 |
4 |
5 |
4 |
5 |
25 |
|
14 |
2 |
5 |
4 |
6 |
2 |
2 |
21 |
|
15 |
3 |
4 |
3 |
5 |
2 |
3 |
20 |
|
16 |
2 |
3 |
6 |
7 |
4 |
3 |
25 |
AM - adult male, AF - adult female, SAM - sub-adult male, SAF -
sub-adult female, JUV - juvenile, INF - infant (N=492).
Table 2. Percentage of feeding time
spent on each plant species by relocated Rhesus Macaques in Asola-Bhatti
Wildlife Sanctuary in Delhi
|
|
Family |
Scientific name |
Common name |
Parts eaten |
Percentage of time spent (%) |
|
1. |
Salvadoraceae |
Salvadora persica |
Meswak |
Leaf, stem |
0.18 |
|
2. |
Fabaceae |
Pithecellobium dulce |
Jungle Jalebi |
Leaf |
7.08 |
|
Prosopis juliflora |
Kikar/Babul |
Leaf, flower |
16.34 |
||
|
Prosopis cineraria |
Khejri |
Leaf |
11.09 |
||
|
Pongamia pinnata |
Indian Beech |
Leaf |
0.14 |
||
|
Acacia nilotica |
Babul |
Leaf, bark |
9.16 |
||
|
3. |
Myrtaceae |
Psidium guajava |
Guava |
Fruit |
0.09 |
|
Syzygium cumini |
Jamun |
Leaf, fruit |
0.03 |
||
|
4. |
Moraceae |
Ficus benjamina |
Fig Tree |
Leaf |
6.7 |
|
Ficus racemosa |
Fig Tree |
Fruit |
0.04 |
||
|
Ficus benghalensis |
Banyan Fig |
Leaf, bark |
1.07 |
||
|
5. |
Euphorbiaceae |
Sapium sebiferum |
Chineese Tallow Tree |
Flower |
0.89 |
|
6. |
Carisseae |
Carissa opaca |
Wild Karonda |
Fruit |
1.08 |
|
7. |
Malvaceae |
Hibiscus ovalifolius |
Roselle |
Flower |
0.47 |
|
8. |
Amaranthaceae |
Alternanthera sp. |
Joyweed |
Leaf |
0.71 |
|
9. |
Poaceae |
Dendrocalamus strictus |
Bamboo |
Leaf |
4.59 |
|
Eleusine indica |
Indian Goosegrass |
Leaf |
0.7 |
||
|
10. |
Legumes |
Cassia fistula |
Amaltas |
Leaf |
0.81 |
|
11. |
Zygophyllaceae |
Balanites aegyptiaca |
Desert Date (Hingot) |
Leaf |
0.59 |
|
12. |
Rhamnaceae |
Zizyphus mauritiana |
Ber |
Leaf |
0.38 |
|
Zizyphus oenoplia |
Ber |
Leaf |
0.29 |
||
|
Zizyphus sp. |
Ber |
Leaf |
0.37 |
||
|
13. |
Cleomaceae |
Cleome viscosa |
Asian Spider Flower |
Leaf |
0.11 |
|
14. |
Capparaceae |
Capparis sepiaria |
Wild Caper Bush |
Leaf |
0.57 |
|
15. |
Rutaceae |
Citrus sp. |
Nimbu |
Leaf |
1.39 |
|
16. |
Apocynaceae |
Calotropis procera |
Rubber Bush |
Leaf |
3.81 |
|
17. |
Meliaceae |
Azadirecta indica |
Neem |
Leaf |
7.56 |
|
18. |
Moringaceae |
Moringa oleifera |
Drumstick Tree |
Fruit |
3.43 |
|
19. |
Combretaceae |
Terminalia arjuna |
Arjun |
Bark |
7.01 |
|
20. |
Solanaceae |
Datura innoxia |
Datura |
Leaf, flower |
7.15 |
|
21. |
Verbenaceae |
Lantana camera |
Sage Tree |
Leaf |
3.68 |
Table 3. Food categories, average time
spent based on seasonal variation, day length of consuming food categories, and
average number of participants at a time during feeding activity
|
Food categories |
Average time spent (%) |
Day length (hours) |
Average number of participants |
|||
|
Summer |
Monsoon |
Winter |
Mean±SE |
|||
|
Plant species |
12.45 |
17.68 |
9.75 |
13.29±2.32 |
5 |
9 |
|
Supplementary food |
53 |
54.32 |
43.25 |
50.19±3.49 |
9 |
32 |
|
Anthropogenic food |
16.55 |
17 |
21 |
18.18±1.41 |
2 |
11 |
|
Provisioned food |
18 |
11 |
26 |
18.34±4.33 |
4 |
29 |