Journal of
Threatened Taxa | www.threatenedtaxa.org | 26 December 2018 | 10(15):
12933–12939
Copulatory behavior of the Jaguar Panthera onca (Mammalia: Carnivora:
Felidae)
Pedro Nacib Jorge-Neto
1, Cristiane Schilbach
Pizzutto 2, Gediendson
Ribeiro de Araujo 3,
Thyara de Deco-Souza 4, Leanes Cruz da Silva 5, Jorge Aparecido Salomão Jr. 6
& Hernan Baldassare 7
1,2,6 Department of Animal Reproduction,
Faculty of Veterinary Medicine and Animal Science, University of São Paulo
(USP) / Av. Prof. Dr. Orlando Marques de Paiva, 87 - Cidade Universitária, São Paulo /
SP, 05508-270, Brazil
3,4 Faculty of Veterinary Medicine and Animal Science, Federal University
of Mato Grosso do Sul (UFMS) / Rua Senador Filinto Muller, 2443 - Vila Ipiranga Campo Grande / MS,
79070-900, Brazil
5 Federal University of Viçosa (UFV) / Avenida Peter
Henry Rolfs, s/n - Campus Universitário, Viçosa / MG, 36570-900, Brazil
7 Department of Animal Science, Faculty of
Agricultural and Environmental Sciences, McGill University, 21111 Lakeshore
Rd., Ste. Anne de Bellevue, Quebec H9X 3V9, Canada
1 pepovet@usp.br,
2 cspizzutto@yahoo.com.br (corresponding author), 3 gediendson@gmail.com,
4 thyara.araujo@ufms.br, 5 leanes.c.s@gmail.com, 6
jorgesalomaojr@usp.br, 7 hernanbalda@gmail.com
Abstract: The relevance of the Jaguar in Brazilian fauna is incompatible with
the lack of literature regarding its reproductive behavior, showing that
research in this area should be intensified.
The knowledge of its basic reproductive behavior is extremely important
for understanding the fertility factors of the species and the role it plays in
its ecosystem. In this study, we
analyzed 210 sequences of sexual behaviors of Jaguars Panthera
onca starting from proceptivity
of the female and ending with copulation; this sequence is called the copulatory behavior.
Behavioral sequences were filmed, and the observed behaviors were
analyzed and recorded including occurrence frequency. Different behaviors were observed in
association with two types of copulation, it was understood that copulation
occurs with and without penile penetration.
The information found in the present study is valuable for the
reproductive management of Jaguars.
Keywords: Copulation, receptiveness,
reproduction, sexual behavior.
Abstract / Resumo: Toda a relevância que
onça pintada tem para a fauna brasileira é incompatível com a ausência de literatura sobre seu comportamento reprodutivo, mostrando que precisamos cada vez mais
intensificar as pesquisas nesta esfera. O conhecimento dos comportamentos reprodutivos básicos é extremamente importante para o entendimento da fecundidade de uma espécie e todo o papel que ela
representa dentro do seu ecossistema. Neste trabalho analisamos 210 sequências de comportamentos sócio-sexuais de onça pintada (Panthera
onca) a partir da proceptividade da fêmea até a finalização da cópula; a esta
sequência chamados de comportamento copulatório. As sequências comportamentais
foram registradas por filmagens e os comportamentos observados foram registrados por frequência de ocorrência. Diferentes comportamentos foram observados em associação com
dois tipos de cópulas, o que nos permitiu concluir
que a cópula ocorre com e sem penetração peniana. As informações encontradas
serão de grande valia para o manejo
reprodutivo da onça pintada.
doi: https://doi.org/10.11609/jott.4218.10.15.12933-12939
Editor: Caroline Leuchtenberger, Instituto Federal de Educação, Farroupilha, Brazil. Date of publication: 26 December 2018
(online & print)
Manuscript details: Ms # 4218 |
Received 24 April 2018 | Final received 198 December 2018 | Finally accepted 20
December 2018
Citation: Jorge-Neto, P.N., C.S. Pizzutto,
G.R. de Araujo, T. de Deco-Souza, L.C. da Silva, J.A.
Salomão Jr. & H. Baldassare
(2018). Copulatory behavior of the Jaguar Panthera
onca (Mammalia: Carnivora:
Felidae). Journal of Threatened Taxa 10(15): 12933–12939; https://doi.org/10.11609/jott.4218.10.15.12933-12939
Copyright: © Jorge-Neto et al. 2018. Creative Commons Attribution 4.0
International License. JoTT allows
unrestricted use of this article in any medium, reproduction and distribution
by providing adequate credit to the authors and the source of publication.
Funding: This study was financed in part
by the Coordenação de Aperfeiçoamento
de Pessoal de Nível
Superior - Brasil (CAPES) - Finance Code 001.
Competing interests: The authors declare no competing interests.
Ethics
statement: This study has been
approved by the Ethics Committee of the School of Veterinary
Medicine and Animal Science, University of São Paulo, under protocol
number 2072180118.
Author
Details: Pedro
Nacib Jorge Neto, DVM, MBA. Actually master’s degree student (PPGRA-FMVZ / USP) and
Technical-Commercial Director of IMV Technologies Brazil. Member of REPROCON research group. Cristiane Schilbach Pizzutto, DVM, MSc, PhD, Postdoc. Professor at FMVZ/USP. Chairman of the
Animal Welfare Committee of CRMV - SP. Member of the International
Environmental Enrichment Conference Committee and of REPROCON research group. Gediendson Ribeiro de Araujo, DVM, MSc, PhD, Postdoc. Veterinarian
at UFMS. Expert in scientific capture of free-living
felids. Member of REPROCON research group. Thyara de Deco-Souza, DVM,
MSc, PhD. Professor at FAMEZ/UFMS. Expert in morphophysiology of reproduction of wild and domestic
animals. Member of REPROCON research group. Leanes Cruz da Silva, DVM, MSc, PhD. Expert in
carnivore conservation. Jorge Aparecido Salomão Jr.,
DVM. Actually master’s degree student (PPGRA-FMVZ / USP).
Works with clinic and surgery of wild and exotic animals in private clinics. Member of REPROCON research group. Hernan Baldassarre, DVM,
PhD. Professor at McGill University (Canada). Expert in small
ruminants reproduction. Global consultant for animal
breeding companies. Member of REPROCON research group.
Author Contribution:
PNJN, CSP, GRA, TDS and HB were responsible
for wrote and review the manuscript; LCS and JASJr
were responsible for collect of the data and analyze of the films.
Acknowledgments: We would like to thank CAPES by post-doctoral fellowship to Dr. Gediendson Ribeiro Araújo and master
fellowship to Pedro Nacib Jorge Neto;
Peter Crawshaw Rescue Center,
Pantanal and all its employees for the support and
the realization of this study; Pedro Busana for the
magnificent drawings.
Introduction
The
Jaguar Panthera onca
is one of the most important top predators among Brazilian fauna. This animal’s
population is decreasing each year due to anthropogenic action, such as
fragmentation of its habitat, farming, hunting and slaughter (Cullen et al.
2016; Olsoy et al. 2016; Espinosa et al. 2018).
Today, this species is considered as Near Threatened by the IUCN Red List
(Quigley et al. 2017) and many research projects involving environmental
education, conservation and reproduction with the species have been developed
in Brazil, such as NEX No Extinction (Corumbá de Goiás - GO), Projeto Onças do Rio Negro (Aquidauana -
MS), Projeto Onçafari
(Miranda - MS), Pantera Brasil
(Poconé - MT) and Projeto
Amigo da Onça (Parque Nacional Boqueirão da Onça - BA), in the attempt to minimize population decrease
and negative consequences to Brazilian
biodiversity (Tortato et al. 2017; Araujo et al. 2018; Diniz et al.
2018).
Basic
information regarding the reproductive behavior of Jaguars is scarce, although
this issue is of extreme importance for the development of assisted
reproduction and conservation actions.
According to Holt et al. (2003) and Owen et al. (2010), reproductive
behaviors and subjacent mechanisms associated with reproductive success are
particularly important, because fitness is fundamentally a fertility
function. For these authors, the study
of reproduction is, therefore, crucial for the conservation of species,
populations and, indirectly, for the vitality of the entire ecosystem.
Ovulation
in this species is usually induced through coitus (Wildt
et al. 1979), however, recent studies involving captive females (Gonzalez et
al. 2017) reported luteal activity in non-pregnant females that were allocated
in individual enclosures, suggesting that spontaneous ovulation occurs
occasionally. Interestingly, through
hormonal analysis of fecal steroids, Barnes et al. (2016) reported
spontaneous ovulations in females housed with males and non-occurrence of
spontaneous ovulation in females maintained without the presence of males,
demonstrating that the Jaguar is a polyestrous species with induced ovulation.
Stehlik (1971)
reported copulatory behavior of the Jaguar at Ostrava Zoo that was briefly described later
in 1976 by Lanier and Dewsbury. These
authors studied and technically described the copulatory
behavior of four Panthera species (P. pardus, P. uncia, P.
tigris and P. onca),
concluding that behavioral patterns were qualitatively similar among them, with
little variation; however, no study with greater description of such behavior
was conducted for the Jaguar.
Limited
information regarding copulatory behavior of Jaguars may be justified due
to their solitary habits, where
interaction with partners is done only during reproductive periods,
which makes behavioral studies very difficult with wild animals. On the other hand, the majority of Jaguars
kept in zoos and Brazilian rescue centers, present particularities that prevent
proper studies with this species, such as animals being too old, castrated,
treated with contraceptives or without a reproductive history. One observation of opportunistic courtship behavior,
in Pantanal, showed the female’s receptiveness and
how the male marks the territory before copulatory
behavior (Leuchtenberger et al. 2009).
In
light of this lack of information concerning the copulatory
behavior of Jaguars and its crucial importance for the development of
reproductive strategies and assisted reproduction projects, the objective of
this study was to analyze a substantial number of sexual interactions of one
adult jaguar couple and thereby describe and characterize the reproductive
behavior of the captive jaguar.
Material and Methods
We monitored a couple of captive adult Jaguars
in the Peter Crawshaw Rescue Center, in southern Pantanal of Brazil.
The male Jaguar was four years old and vasectomized; the female was
eight years old at the time of the study.
The animals’ enclosure measured 39m in width and 49m in length. Animals were paired during the entire year
and were monitored through cameras Intelbras VDH 5040
VF G2, 24 hours.
A total of 210 films recorded four
consecutive natural estrus.
Sexual behaviors were registered using the continuous focal method
(Martin & Bateson 2007). Moreover,
for this study, the proceptivity of the female until
the effectiveness of copulation was considered.
This sequence of behavioral events was entitled copulatory
behavior. A large portion of the
methodology for behavioral evaluation was adapted from the classification
described by Lanier & Dewsbury (1976), and can be found in Table 1.
Statistical
Analyses
Each copulatory behavior sequence, which encompassed the period
between female proceptivity and the end of
copulation, was considered as one film event.
In each film, each behavior (as specified in Table 1) was considered as
one registered occurrence. At the end of
each film, a quantitative analysis of occurrence frequency (%) was performed
for each behavior presented in relation to the total occurrences in all filmed
sequences. Further analysis of copulatory behaviors between event
ending with vs. without penile penetration was conducted by contingency
analysis and Fisher’s exact test. Differences were considered to be
statistically significant at the 95% confidence level (P<0.05).
Results
The
duration of each estrus period based on female receptivity were nine, eight,
eight and 10 days for the four estrus cycles evaluated and the estrus-to-estrus
interval was 34, 39 and 30 days. The
visualization and consequent confirmation of penile introduction during
copulation occurred in 10 episodes. All
behavior sequences observed during female receptivity (210 events) until the
end of the male´s pelvic movement behavior were similar. The analysis of the male’s pelvic movement
behavior showed two different behavioral sequences, characterized as copulatory behavior with penile penetration (122 events)
and copulatory behavior without penile penetration
(88 events).
In the
sequence of copulatory behavior without penile
penetration, the male left the female after finishing his pelvic movements in
42% of the sequences observed. In turn,
in the sequence with penile penetration, additional copulatory
behaviors were observed from this moment on, such as the male biting or licking
the female’s nape, and female rocking and rolling in lateral-dorsal decubitus
in 58% of observations. The frequencies
of sexual behavior occurrences that involved both Jaguars’ copulatory
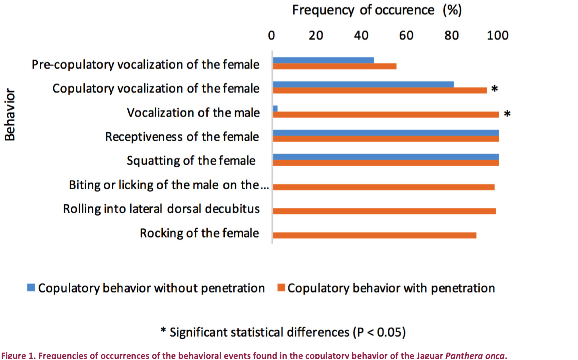
behavior sequences can be observed in Figure 1:
The positioning of the female
(squatting) was verified in 100% of our observations;
Male vocalization was observed
in only 1/88 copulatory events without penetration
(P<0.001);
High copulation frequency could
be partly explained by the fact that penile penetration occurred only in 42% of
the mounts;
Female vocalization during proceptivity occurred at practically the same frequency
preceding copulations with and without penile penetration (55% and 45%,
respectively; P>0.05);
During copulation, female
vocalization was more frequent when there was penetration (95.1% vs. 79.6%,
P<0.01);
When
penile penetration could be confirmed, the copulatory
behavior presented the following sequence of events:
Proceptivity of the
female with tail movement and presentation to the male (Figure 2.1)
The female lies in ventral
decubitus, deviating the tail to the side and exposing the anal-genital region
to the male; the thoracic limbs of the female are, in general, elongated and
the pelvic limbs are flexed next to the body (Figure 2.2)
The male mounts from the back and on top of the female, keeping her between
his front paws. He then approximates his
genital region to the female’s, squatting with the
pelvic limbs
The female
deviates the tail and the male initiates pelvic impulse – the penis is
introduced – the male bites/licks the female’s nape three or four times (Figure
2.3) – the female may or may not emit a low growling – the male roars,
presumptively indicating ejaculation
Female rocking – rolling into lateral dorsal decubitus
(Figure 2.4).
Discussion
The
results found in this study represent the first complete descriptions of the copulatory behavior specific to the Jaguar Panthera onca since
the 1970s, which is information of great relevance for studies involving
biology and even reproductive biotechnologies.
In this context, the understanding of reproductive behaviors and
aptitude of any species is critical for the understanding fertility (Owen et
al. 2010).
One
opportunist observation (Leuchtenberger et al. 2009)
and previous studies from Lanier & Dewsbury (1976) and Stehlik
(1971) reported rudimentary information regarding the reproductive behavior of
the genus Panthera, but only part of these
reports involving copulation in the genus Panthera
can be considered specifically for the Jaguar.
By initiating observations
from the moment the female shows herself
as proceptive to the male, it was clear that the
percentage of approach from the male to the female corresponded exactly to the
percentage of female proceptivity, thereby indicating
that female signalization for possible receptiveness is highly effective in
triggering male attractiveness and initiation of courting.
From
the moment when the female was receptive to the male and male pelvic movements
began, in 42% of the sequences observed, the male finished the copulatory behavior and left the female. This supported the conclusion that this would
be a copulatory behavior without penile
penetration. In 58% of observations, the
male remained in the act of copulation and started biting or licking the
female’s nape. In turn, the female started
rocking and rolling in lateral-dorsal decubitus. In this situation, we concluded that penile
penetration occurred. When penile
penetration occurred, the male vocalized in 100% of the events before lightly
biting or licking the female’s nape, as described by Hancock (2000) for
leopards and by Lanier & Dewsbury (1976) for Jaguars. In contrast, male vocalization was observed
in only 1/88 copulatory events without penetration
(P<0.001). These findings are
consistent with the study by Lanier & Dewsbury (1976), who described that
the males’ roar during copulation was verified in every observation of
copulation with penile penetration, signalizing success in ejaculation.
It is
interesting to point out that the elevated number of copulations in felid
species, according to Wildt et al. (2010), has been
proposed as a method to induce multiple ovulations among females, and also, in
species with high incidence of teratospermia, to
ensure deposition of an adequate amount of normal sperm in the vagina, thereby
increasing the chance of pregnancy.
According
to other comparative in situ and ex situ studies, when compared with others
wild felid species such as Cheetahs (Crosier et al. 2009), Clouded Leopards (Wildt et al. 1986), and Oncillas
(Swanson & Brown 2004) teratospermia (>60% of
defective spermatozoids) is not common in the Jaguar (Morato
et al. 2001; Araujo et al. 2018; Gonzales et al.
2017) . Nevertheless, multiple copulations were observed in the present
study. High copulation frequency could
be partly explained by the fact that penile penetration occurred only in 42% of
the mounts, so the male continued attempting to mount until completing
ejaculation. Nevertheless, we believe
that future studies must be conducted in a format allowing the confirmation of
semen deposition in the vagina.
Every
positioning of the female, described by Lanier & Dewsbury (1976), such as
squatting (elongated anterior limbs and flexed posterior limbs next to the
body) was verified in 100% of our observations, both for copulations considered
as “with” and “without” penetration.
This suggests that the success of penile penetration is not related to
female posture, because the female was found in the same position in both
situations.
Female
vocalization during proceptivity did not influence
the result of copulation since it occurred at practically the same frequency
preceding copulations with and without penile penetration. During copulation, however, female
vocalization was more frequent when there was penetration. Only when copulation involved penile
penetration was the female rocking followed by rolling into
lateral dorsal decubitus. These
findings are consistent with observations by Stehlik
(1971) but in disagreement with report by Lanier & Dewsbury (1976), who did
not describe female rolling
into lateral dorsal decubitus as characteristic behavior after
copulation.
Conclusions
- Basic information regarding the reproductive behavior of Jaguars is of extreme importance for the
management, development of assisted reproduction and conservation projects.
- Copulatory behavior of the Jaguar was described in a
qualitative manner starting from first signs of female proceptivity
and until the finalization of copulation.
- We found that close to half of the
copulation events of Panthera onca might occur without penile penetration and, in
these cases, ejaculation is believed not to occur.
-
Numerous copulations occured during female
estrous. Consistent with thoughts
reported by others, we believe this behavior may be necessary to promote
multiple ovulations and to ensure sufficient number of successful penile
penetrations with ejaculation, thereby ensuring proper
numbers of normal fertilizing sperm are deposited in the vagina.
References
Araujo, G.R.,
T.A.R. Paula, T. Deco-Souza, R.G. Morato, L.C.F. Bergo, L.C. Silva, D.S. Costa & C. Braud
(2018). Comparision of sêmen samples collect from wild and captive Jaguars (Panthera onca) by
urethral catheterization after pharmacological induction. Animal
Reproduction Science 195: 1–7;
https://doi.org/10.1016/j.anireprosci.2017.12.019
Barnes,
S.A., J.A. Teare, S. Staaden,
L. Metrione & L.M. Penfold
(2016). Characterization
and manipulations of reproductive cycles in the Jaguar (Panthera
onca). General and Comparative Endocrinolology 225: 95-103; https://10.1016/j.ygcen.2015.09.012
Crosier,
A.E., J.N. Henghali, J. Howard, B.S. Pukazhenthi, K.A. Terrell, L.L. Marker & D.E. Wildt (2009). Improved quality of
cryopreserved Cheetah (Acinonyx jubatus) spermatozoa after centrifugation through accudenz. Jounal of Andrology 30: 298–308; https://10.2164/jandrol.108.006239
Cullen, L. Jr., J.C. Stanton, F.
Lima, A. Uezu, M.L.L. Perilli
& H.R. Akçakaya (2016). Implications of fine-grained
habitat fragmentation and road mortality for Jaguar conservations in the
Atlantic forest, Brazil. PLoS One
11(12): e0167372; https://doi.org/10.1371/journal.pone.0167372
Diniz, M.F.,
R.B. Machado, A.A. Bispo & D. Brito
(2018). Identifying key sites for connecting Jaguar populations in
the Brazilian Atlantic Forest. Animal Conservation 21: 201-210; https://10.1111/acv.12367
Espinosa, S., G. Celis
& L.C. Branch (2018). When roads appear jaguars
decline: Increased access to an Amazonian wilderness area reduces potential for
Jaguar conservation. PLoS One 13(1): e0189740; https://doi.org/10.1371/journal.pone.0189740
Gonzalez, S.J., J.G. Howard, J.
Brown, H. Grajales, J. Pinzón,
H. Monsalve, M.A. Moreno & C.J. Escobar (2017). Reproductive analysis of male
and female captive Jaguars (Panthera onca) in a Colombian zoological park. Theriogenology 89: 192-200; https://10.1016/j.theriogenology.2016.09.049
Hancock, D. (2000). A time with leopards.
Black Eagle Publishing, Cape Town, South Africa.
Holt,
W.V., A.R. Pickard, J.C. Rodger & D.E. Wildt
(2003). Reproductive Science and Integrated Conservation. Cambridge University Press, Cambridge, United Kingdom, 20pp.
Lanier,
D.L. & D.A. Dewsbury (1976). A
quantitative study of copulatory behaviour
of large felidae. Behavioural
Processes 1(4): 327–333; https://doi.org/10.1016/0376-6357(76)90014-0
Leuchtenberger, C.,
P. Carwshaw, G. Mourão
& C. Lehn (2009). Courtship behavior by Jaguars
in the Pantanal of Mato Grosso do Sul. Natureza
& Conservação 7(1): 218–222.
Martin, P. & P. Bateson (2007). Measuring
Behavior: An Introductory Guide. United States of America:
Cambridge University Press, 176pp.
Morato, R.G.,
V.A. Conforti, F.C. Azevedo,
A.T. Jacomo, L. Silveira,
D. Sana, A.L. Nunes, M.A. Guimaraes
& R.C. Barnabe (2001). Comparative analyses of semen and endocrine characteristics of free-living
versus captive Jaguars (Panthera onca). Reproduction
122(5): 745–751.
Olsoy, P.J.,
K.A. Zeller, J.A. Hicke, H.B. Quigley, A.R. Rabinowitz & D.H. Thornton (2016).
Quantifying the effects of deforestation and fragmentation on a range-wide
conservation plan for Jaguars. Biological Conservation 203: 8–16; https://doi.org/10.1016/j.biocon.2016.08.037
Owen, C., S. Niemann
& R. Slotow (2010). Copulatory
parameters and reproductive success of wild Leopards in South Africa. Journal
of Mammology 91(5): 1178–1187; https://doi.org/10.1644/09-MAMM-A-256.1
Quigley, H., Foster, R., Petracca,
L., Payan, E., Salom, R.
& Harmsen, B. (2017). Panthera
onca (errata version published
in 2018). The IUCN Red List of Threatened Species 2017:
e.T15953A123791436. Downloaded on 20 December
2018; https://doi.org/10.2305/IUCN.UK.2017-3.RLTS.T15953A50658693.en
Stehlik, J.
(1971). Breeding Jaguars at Ostrava Zoo. International Zoo
Yearbook 11(1): 116–118; https://doi.org/10.1111/j.1748-1090.1971.tb01871.x
Swanson, W.F. & J.L. Brown (2004). International training programs in reproductive sciences for
conservation of Latin American felids. Animal Reproduction Science
82–83: 21–34; https://doi.org/10.1016/j.anireprosci.2004.05.008
Tortato, F.R.,
T.J. Izzo, R. Hoogesteijn
& C.A. Peres (2017). The numbers of the beast: Valuation of
Jaguar (Panthera onca)
tourism and cattle depradation in the Brazilian Pantanal. Global Ecology and Conservation 11:
106–114; https://doi.org/10.1016/j.gecco.2017.05.003
Wildt, D.E.,
C.C. Platz, P.K. Chakraborty
& S.W. Seager (1979). Oestrous and
ovarian activity in a female Jaguar (Panthera
onca). Journal of Reproduction and
Fertility 56(2): 555–558; https://doi.org/10.1530/jrf.0.0560555
Wildt, D.E.,
J.G. Howard, L.L. Hall & M. Bush (1986). Reproductive physiology
of the clouded leopard: I. Electroejaculates contain
high proportions of pleiomorphic spermatozoa
throughout the year. Biology Reproduction 34: 937–47; https://doi.org/10.1095/biolreprod34.5.937
Wildt,
D.E., W.F. Swanson, J.L. Brown, A. Sliwa, & A.
Vargas (2010). Felids ex situ for managed programmes,
research, and species recovery, pp. 217–235. In: MacDonald, D.W. & A. Loveridge (eds.). Biology and
Conservation of Wild Felids. Oxford: Oxford University Press.
Table 1. Behavioral
catalog used for the description of the copulatory behavior of the couple
of Jaguars Panthera onca
at the Peter Crawshaw Rescue Center,
in southern Pantanal
|
Sexual behavior |
Definition |
|
Pre-copulatory vocalization of
the female |
When the female vocalizes during proceptiveness |
|
Copulatory
vocalization of the female |
When the female vocalizes during the copula |
|
Vocalization of the male |
When the male vocalizes during the copula |
|
Attractiveness of the male |
When the male approaches the female and initiates the
interaction that may lead to the copula |
|
Proceptiveness of
the female |
When the female requests the male, approaching and turning
to him, with presentation of the anal-genital region; |
|
Receptiveness of the female |
When the female accepts mount from the male |
|
Squatting of the female |
When the female squats in ventral decubitus, in sexual
receptiveness posture |
|
Biting or licking of the male on the females nape |
When the male licks or bites the females nape during the
copula |
|
Rocking of the female |
When the female, after the copula, turns around and hits
the male with one paw |
|
Rolling of the female |
When the female rolls into lateral dorsal decubitus after
the copula |
|
Copula without penial
introduction |
When the pelvic movement of the male occurs during the
mount on the female, however, without introduction of the penis |
|
Copula with penial introduction |
When the pelvic movement of the male occurs during the
mount on the female, followed by the introduction of the penis. |