Journal of
Threatened Taxa | www.threatenedtaxa.org | 26 October 2018 | 10(11):
12538–12551
Pollination ecology of Synedrella
nodiflora (L.) Gaertn. (Asteraceae)
B. Usharani
1 & A.J. Solomon Raju
2
1,2 Department of Environmental Sciences,
Andhra University, Visakhapatnam, Andhra Pradesh 530003, India
1 ushabudumajji@gmail.com, 2 solomonraju@gmail.com
(corresponding author)
Abstract:
Synedrella nodiflora (L.) Gaertn
grows almost throughout the year if the soil is damp. It produces heterogamous capitula
with female ray florets anthesing on the first day
and bisexual disc florets anthesing on the next three
consecutive days. Disc florets are dichogamous, herkogamous,
self-compatible, self-pollinating (vector-mediated) and display secondary
pollen presentation through an intermediate form of brush mechanism. Ray and disc florets exhibit facultative xenogamy.
Butterflies are principal pollinators while bees,
wasps and flies are supplementary pollinators.
Thrips Microcephalothrips
abdominalis uses the florets as breeding and
feeding sites; the feeding activity effects pollination. The ray and disc florets produce cypselas; the cypselas produced
by ray florets are heavier, elliptical, membranous with upwardly-pointing teeth
along the margins and two short terminal awns while those produced by disc
florets are lighter, cylindrical and tangentially compressed with 2 or 3 stiff
terminal divaricate awns. Seed dispersal
is polychorous and represented by anemochory,
anthropochory, zoochory and
ombrohydrochory.
Cypselas of ray florets disperse to short
distances and germinate under specific germination conditions either at
parental sites or in similar habitats while those of disc florets disperse
farther away from parental sites and germinate readily under a wide range of
conditions. Therefore, bimorphic cypselas with different
germination abilities enable the plant to grow as a widespread weed but not as
an invasive weed.
Keywords: Bimorphiccypselas,
entomophily, polychory, secondary pollen
presentation, Synedrella nodiflora, thripsophily.
doi: https://doi.org/10.11609/jott.4008.10.11.12538-12551
Editor: Kannan C.S. Warrier,
Institute of Forest Genetics and Tree Breeding, Coimbatore, India. Date
of publication: 26 October 2018 (online & print)
Manuscript details: Ms
# 4008 | Received 13 January 2018 | Final received 14 September 2018 | Finally
accepted 26 September 2018
Citation: Usharani, B. & A.J.S. Raju (2018). Pollination
ecology of Synedrella nodiflora
(L.) Gaertn. (Asteraceae). Journal
of Threatened Taxa 10(11): 12538–12551;
https://doi.org/10.11609/jott.4008.10.11.12538-12551
Copyright: © Usharani & Raju 2018. Creative Commons
Attribution 4.0 International License. JoTT
allows unrestricted use of this article in any medium, reproduction and
distribution by providing adequate credit to the authors and the source of
publication.
Funding: Self-funded.
Competing interests: The authors declare no competing interests.
Author
Details: Ms.
B. Usharani
is a PhD student in the Department of Environmental Sciences, Andhra
University. Recently, she has been awarded PhD under the guidance of Prof. A.J.
Solomon Raju.
She has published two research papers on pollination ecology. Prof.
A.J. Solomon Raju is working in the Department
of Environmental Sciences, Andhra University, Visakhapatnam.
He is the recipient of several national and international awards. He has more
than 300 research papers in international and national Journals. He is on the editorial board of several
international journals. He visited USA,
Canada, UK, Spain, Brazil, Paraguay, Mexico, Italy, Malaysia, Thailand,
Ethiopia, Tanzania, Hong Kong, China and Australia.
Author
Contribution: Both the
authors contributed equally.
Acknowledgements: We thank the Andhra University, Visakhapatnam, for providing physical
facilities to carry out this research work.
INTRODUCTION
Synedrella is a monotypic genus with only a single species, S. nodiflora. The
generic name is derived from the Greek word “synedros”
meaning small flowers seated together while the species name indicates the
presence of flower clusters around the nodes in the upper parts of the plant (Davidse et al. 2015).
It is native to tropical America and from there it has become widespread
throughout the warmer regions of the world (Holm et al. 1997). It is spread
throughout the Southeast Asian region, found in the plains of India, in the Andamans and West Africa.
It is also found in Bangladesh, Japan, Spain, China and England (Chauhan & Johnson 2009). The plant is used as feed for livestock and
in traditional medicine to treat various health problems in Ghana, Nigeria,
Malaysia and Indonesia (Burkill 1985; Idu & Onyibe 2007). This plant displays floral features
which are characteristic of thripsophily (Ananthakrishnan 1993).
Further, it is reported that this plant with small and less attractive
heterogamous heads is inhabited only by one terebrantian
thrip, Microcephalotrhips
abdominalis which is able to move freely within
the capitulum and also fly to other capitula of the same plant or other nearby plants. As a result, they effect
both self and cross-pollination (Ananthakrishnan et
al. 1981).
Kissmann & Groth (1992) reported that Synedrella nodiflora
produces dimorphic cypselas. Rocha (1996) stated that the morphological
differences between these cypselas influence
dispersion strategies directly. Bradford
(1990) stated that both the cypselas of S. nodiflora exhibit the same pattern of imbibition rates
indicating no relationship to the germination rate or differences in the fruit
coat permeability. Brandel
(2004) reported that the germination responses between these cypsela types are
not so marked because of a lack of deep innate dormancy. However, they show some differences in germination
rates depending on their environment perception. The state of information available on
pollination and seed ecology is insufficient to understand the pollination and
propagation abilities of S. nodiflora as a
widespread weed across tropical latitudes.
In this context, the present study was contemplated to investigate the
following objectives: flowering phenology, floral biology, pollination
mechanism, pollinators, sexual system and seed dispersal ecology. This information presented in this paper enables
us to understand the studied aspects of S. nodiflora.
MATERIALS AND METHODS
Populations of Synedrella nodiflora growing in Visakhapatnam region (17.6860N
& 83.2180E ) in Andhra Pradesh State,
India were used for the study from 2015–2017.
Observations regarding the organization of inflorescences, the spatial
positioning of flowers, and their position on the plant were made since these
features are regarded as important for effecting pollination by foragers. The life time of
individuals of two floret types was recorded by marking twenty just open
florets each and following them until fall off.
Anthesis was initially recorded by observing
ten marked mature capitula in the field. Later, the observations were repeated five
times on different days; on each day we observed twenty marked mature capitula in order to provide an accurate anthesis schedule.
Twenty mature disc florets were followed for recording the time of
anther dehiscence. The presentation
pattern of pollen was also investigated by recording how anthers dehisced and
confirmed by observing the anthers under a 10x hand lens. The details of flower morphology such as
flower sex, shape, size, colour, odour, sepals, petals, stamens and ovary were
described.
Twenty
mature but undehisced anthers from disc florets were
collected from five randomly chosen plants and placed in a petri dish. The
pollen output per anther/disc floret and pollen-ovule ratio was calculated
using the protocol given by Cruden (1977). Individual volumes of nectar were recorded
for 25 ray and 25 disc florets and then the average volume of nectar per each
floret type was determined and expressed in µl.
The capitula used for this purpose were bagged
at the mature bud stage, opened after anthesis and
squeezed the nectar from each floret into a micropipette to measure the volume
of nectar. Based on nectar volume in
individual ray and disc florets, the total volume of nectar secreted in a capitulum was calculated.
Similarly, the nectar sugar concentration at the capitulum
level was determined using a Hand Sugar Refractometer
(Erma, Japan). Nectar analysis for sugar
types was done as per the Paper Chromatography method described in Dafni et al. (2005).
The sugar content/floret is expressed as the product of nectar volume and
sugar concentration per unit volume, mg/µl.
This was done by first noting the conversion value for the recorded
sugar concentration on the refractometer scale and
then by multiplying it with the volume of nectar/floret. Table 5.6 given in Dafni
et al. (2005) was followed for recording the conversion value to mg of sugars
present in one µl of nectar. Dinitrosalicylic acid method was followed for the first two
sugar types while Resorcinol method was followed for the last sugar type. The caloric reward of nectar/floret/day was
measured as per the formula given in Heinrich (1975). He assumed that 1 mg of sugar yields 16.74
joules or 4 calories of energy and accordingly he used the formula for
calculating the caloric reward of the nectar.
Nectar volume (µl) X Concentration of nectar (%)
--------------------------------------------------------------- x 16.74
100
Paper chromatography method described in Dafni
et al. (2005) was followed for identifying the amino acid types in the nectar
of ray and disc florets; the amino acid types were the same in both ray and
disc florets. The flower visitors were
collected and identified with the representative specimens available with the
Department of Environmental Sciences, Andhra University, Visakhapatnam. All butterflies were further confirmed by
consulting the books of Kunte (2007) and Gunathilagaraj et al. (1998) while other insects, some to
species level while a few others to genus level only. Thrips were
identified using the key provided by Bhatti (1980)
for Indian thrips.
The insects were observed carefully for 10 hours a day for 15 days in
different months each year during the profuse flowering period. The hourly foraging visits of each species
were recorded on ten different days for which thirty capitula
were selected. The data obtained was
used to calculate the percentage of foraging visits made by each species per
day and also to calculate the percentage of foraging visits of each category of
insects per day. Simultaneously, the insects were observed for their foraging behavior such as mode of approach, landing, probing
behaviour, the type of forage they collected, contact
with essential organs to result in pollination, and inter-plant foraging
activity. The insects were captured from
the capitula during 10:00–12:00 hr
on five different days for pollen analysis in the laboratory. For each insect species, 10 specimens were
captured and the proboscides were separated for examination to record whether
they carried pollen grains or not. They
were washed first in ethyl alcohol and the contents stained with aniline-blue
on a glass slide and then observed under a microscope to count the number of
pollen grains present.
A
sample of capitula was tagged and followed for fruit
set rate of ray and disc florets in open-pollinations and the same sample was
used for noting the duration of fruit maturation. Seed characteristics of ray and disc florets
were carefully examined to note their special adaptations for dispersal modes. Field visits were made to record whether the
seeds germinate immediately after they are dispersed or not. Field observations on seed germination and
seedling formation were made to record the approximate number of generations
produced during the rainy season.
RESULTS
Phenology
The
plant is an erect, branched, ephemeral herb with long internodes and swollen
nodes (Image 1a). The lower parts of the
stems root at the nodes in damp soil conditions. The shallow tap root system is strongly
branched. It grows in humid places,
shaded and nutrient rich soils, crop fields, wastelands, roadsides, lawns and
disturbed areas. The stems are ribbed,
dichotomously branched, sub-angular, smooth to
sparsely hairy. The leaves occur in
opposite pairs, ovate to elliptic, finely hairy with short petioles and joined
by a ridge across the stem. It
propagates only by seed. Individual
plants complete their life cycle within 4–5 months. The seeds germinate and produce new plants as
soon as they are dispersed from the mother plants and in effect, the plants
show vegetative, flowering (Image 1b) and fruiting phases simultaneously in
different habitats. However, the plant
shows prolific growth during the rainy season and displays peak flowering from
October-November.
Flower morphology
A plant
produces several capitula and their numbers vary
depending upon the age, number of branches and nutrient condition of the
soil. But, it produces an average of
22.6±4.6 capitula.
The capitula are sessile and borne either
singly or as groups of 2–8 to nodes in leaf axils; they are
covered by two green herbaceous involucral bracts.
Each capitulum consists of odorless
outer ray florets and central disc florets and each floret
type is enclosed by 3–5 mm long erect bracts. Ray florets vary from 5–8 (7.3±1.31) while
disc florets vary from 11–15 (13.06±1.50); they are produced in the ratio of
1:2. The calyx is reduced to pappus of 2–3 barbed and strong bristles in both types of
florets. The corolla is tubate, zygomorphic, yellow and tipped with one ligulate lobe in ray florets and five actinomorphic lobes
in disc florets. The ray floret is
8.1±0.7 mm long and 2.0±0.3 mm wide while disc floret is 8.2±0.6 mm long and
1.2±0.3 mm wide. The ray florets lack
stamens but have well developed pistil with ovary and
style which is terminated into two smooth stylar arms
(Image 1h,i). The disc florets have four
stamens with yellow filaments and dark brown anthers. The stamens are epipetalous and anthers are dithecous, fertile, united and cohered forming a hollow
cylinder representing syngenesious condition (Image
1m). The anther has
its connective prolonged into a hood and tend to separate when the
flower withers. The pistil is well
developed with ovary and long style which is
terminated into two linear-lanceolate stylar arms covered abaxially at
the base with sweeping hairs. A nectariferous disc is present at the base of the style
inside the corolla tube. The style with
its aligned arms extend beyond the height of anthers (Image 1j,k,n); the stylar arms diverge and
curve inwards exposing the hidden stigmatic surfaces and finally over-arching
the florets (Image 1l). In both floret
types, the ovary is bicarpellary, syncarpous
and unilocular with one anatropous ovule on basal
placentation. The floral features indicate that ray florets are female and disc
florets are bisexual.
Floral biology
The ray
and disc florets open during early morning from 07:00-09:00 hr
on clear sunny days(Image 1c–g) (Table 1). The florets
open completely on sunny days while they are partially open on rainy days. In a capitulum, the
ray florets open first and all of them open simultaneously; the disc florets
open concentrically inwards from the next day for three successive days with
38% opening on day 1 and 31% each on day 2 and day 3 (Table 1). Individual ray and disc florets take about
three hours to open from mature bud phase.
The disc florets are protandrous with anther
dehiscence taking place during mature bud stage by longitudinal slits. The narrow anthers are united to form a
hollow space into which pollen is liberated.
At the mature bud stage the style with its aligned stylar
arms lies below the anthers, its forked arms are converged at this stage. During and immediately after anthesis, the style grows, elongates and passes through the
anther tube brushing the pollen with its sweeping hairs. At this stage, the inner stigmatic surfaces
are un-receptive and not exposed, the functional situation of which prevents
the occurrence of autogamy. Such a form
of pollen presentation is referred to as “secondary pollen presentation
mechanism” which ensures the pollen availability to insects visiting the capitula on a daily basis.
The style with its aligned branches gradually diverge
in the early hours of the second day; then the inner stigmatic surfaces attain
receptivity and remain so until the end of that day. The stylar arms
curve downwards completely exposing the receptive stigmatic surfaces. Ray florets display stigma receptivity by
diverging the stylar arms day-long
on the day of anthesis only. The pollen grains are bright yellow,
spheroidal, tricolporate, 35.96±0.04
µm in size and echinate. The pollen grains are
134.13±21.16 per anther, 536.53±84.66 per floret and 6,975 per capitulum (Image 1o).
The pollen-ovule ratio is 955:1.
The ray and disc florets are nectariferous. A
ray or disc floret produces 1.3μl of nectar which
rises up as it accumulates in the floret due to the narrow corolla tube. The beginning of nectar secretion coincided
with anthesis in ray florets and with anther
dehiscence in disc florets; its secretion ceased by the evening of day 1 in ray
florets while it ceased by the evening of day 2 coinciding with cessation of
stigma receptivity. A capitulum produces an average volume of 22.4μl of nectar
during its lifespan; the nectar sugar concentration is 27±1.6 % with 0.4mg of
sugar containing 1.6 calories of energy at floret level and 6.67mg sugar
containing 26.68 calories of energy at capitulum
level. The sugar types present in the
nectar include sucrose, glucose and fructose; they are present in that order of
dominance. The nectar contains five
essential amino acids (arginine, histidine, lysine,
tryptophan and threonine) and six non-essential amino acids (cysteine, glycine,
serine, aspartic acid, glutamic acid, hydroxyproline,
alanine, cystine and proline);
they are present in that order of dominance.
The ray and disc florets wither away on the 3rd day and fall
off on the 4th day. The tubate corolla in ray florets and the tubate
corolla and stamens in disc florets gradually fall off following fertilization
and initiation of fruit formation.

Table 1. Anthesis
of disc florets as a function of time in Synedrella
nodiflora
|
Time (h) |
No. of disc
florets anthesed |
||||||
|
Day 1 |
% |
Day 2 |
% |
Day 3 |
% |
Total |
|
|
06:00 |
- |
- |
- |
- |
- |
- |
- |
|
07:00 |
2 |
15 |
1 |
8 |
1 |
8 |
31 |
|
08:00 |
2 |
15 |
2 |
15 |
2 |
15 |
45 |
|
09:00 |
1 |
8 |
1 |
8 |
1 |
8 |
24 |
First, all ray florets open at once on the
first day of anthesis of capitulum. The next day onwards disc florets open for
three consecutive days. Total No. of disc florets per capitulum 13
Table 2. List of insect foragers on Synedrella nodiflora
|
Order |
Family |
Genus |
Species |
Common name |
Forage sought |
|
Hymenoptera |
Apidae |
Trigona |
iridipennis Smith |
Stingless Honey Bee |
Pollen + Nectar |
|
|
|
Ceratina |
sp. |
Small Carpenter Bee |
Pollen + Nectar |
|
|
|
Ceratina |
smaragdula F. |
Small Carpenter Bee |
Pollen + Nectar |
|
|
Anthophoridae |
Xylocopa |
latipes L . |
Large Carpenter Bee |
Nectar |
|
|
Halictidae |
Nomia |
sp. |
Alkali Bee |
Pollen + Nectar |
|
|
|
Lasioglossum |
sp. |
Sweat Bee |
Pollen + Nectar |
|
|
Vespidae |
Rhynchium |
sp. |
Black Potter Wasp |
Nectar |
|
|
Sphecidae |
Ammophila |
sp. |
Digger Wasp |
Nectar |
|
Diptera |
Sarcophagidae |
Sarcophaga |
sp. |
Flesh Fly |
Nectar |
|
|
Syrphidae |
Eristalinus |
sp. |
Hover Fly |
Nectar |
|
Lepidoptera |
Pieridae |
Eurema |
hecabe L. |
Common Grass Yellow |
Nectar |
|
|
|
Leptosia |
nina F. |
Psyche |
Nectar |
|
|
Lycaenidae |
Zizula |
hylax F. |
Tiny Grass Blue |
Nectar |
|
|
|
Pseudozizeeria |
maha Kollar |
Pale Grass Blue |
Nectar |
|
|
|
Zizeeria |
karsandra Moore |
Dark Grass Blue |
Nectar |
|
|
|
Everes |
lacturnus Godart |
Indian Cupid |
Nectar |
|
|
Hesperiidae |
Pelopidas |
mathias F. |
Small Branded Swift |
Nectar |
Pollination mechanism
The ray
florets devoid of stamens act as female and they expose the stigmatic region
prominently by unfolding stylar arms immediately
after anthesis against the ligulate
petal to receive pollen from the foragers on par with disc florets. The disc florets present the stamens and stylar arms at different positions. The anthers dehisce inwardly and discharge
pollen grains into the anther tube during mature bud stage. At this stage, the style lies below the basal
part of the anthers. During and
immediately after anthesis, the style with its
aligned stylar arms elongates within the anther tube
and brushes the pollen out of the anther tube by stylar
hairs called “sweeping hairs” presenting the pollen only on the abaxial surface at the base of stylar
arms (sterile portions). Such a pollen
presentation pattern is indicative of secondary pollen presentation functional
through an intermediate mechanism representative of brush mechanism. The stylar arms are
in closed state on day 1 and the inside stigmatic surfaces are un-receptive, there is no possibility for the occurrence of
autogamy. The staminate
phase ends at 1700 h and the sterile portion of the stylar
arms has residual pollen at that time.
This is followed by the retraction of filaments and the consequent
partial retraction of anthers into the corolla tube. The pistillate
phase starts in the early hours of the second day, the stigmatic surfaces
attain receptivity, gradually diverge and curve downwards within three hours
totally exposing the inner stigmatic surfaces; the stigmatic surfaces remain
receptive until the end of that day. The
brush type pollen presentation mechanism, staminate
phase on day 1 and pistillate phase on day 2 appear
to have evolved to prevent autogamy and promote cross-pollination. However, the anthesis
of disc florets for three consecutive days in the same and different capitula on the same plant facilitates the occurrence of
vector-mediated self-pollination.
Therefore, the secondary pollen presentation mechanism and the sexual
system function do not insulate completely from the occurrence of self-pollination
and hence the flowers set fruit and seed through self- as well as
cross-pollination.
Thrips breeding, feeding and pollination
Thrips
species, Microcephalothrips abdominalis (Thysanoptera: Thripidae) oviposited during
early bud stage of florets of capitula. The larvae emerge from the eggs in synchrony
with anthesis and nectar production in both ray and
disc florets. The centripetal
development of the capitulum was found to provide
continuous availability of pollen and nectar for three days for their
growth. The thrips
were found feeding on pollen and nectar, especially from disc florets. They were dusted with pollen in their upward
and downward movements within the corolla tube of disc florets. The echinate pollen
surface facilitated the thrips to carry 180 to 345
pollen grains on their body setae, wings and legs. The thrips
dispersed the pollen on the stigmatic region of the stylar
arms of both ray and disc florets due to their active movements, rubbing the
abdomen on the stigmatic surface, cleansing of their body parts with their hind
legs and also by their wing combing mechanism.
Since the disc floret is staminate on day 1
and pistillate on day 2, the foraging activity of thrips within that floret does not contribute to
self-pollination but may contribute to self-pollination between disc florets of
the same or different capitula of the same plant.
Further, self-pollination may also take place in ray florets due to deposition
of pollen of disc florets of the same capitulum by thrips. As the plant
occurs as small or large populations, thrips could
fly to migrate to the capitula of other closely
spaced plants and effect cross-pollination.
Insect foraging activity
The capitulum is the unit of attraction for insect foragers.
Within the capitulum, the yellow ligulate
petal of ray florets acts as chief attractant.
The ray and disc florets were foraged by bees, wasps, flies and
butterflies. The butterflies were the
consistent and regular foragers while all others were inconsistent foragers and
also they forage especially during the peak flowering season. The bees were Trigona
iridipennis (Image 2a,b), Ceratina
sp. (Image 2d), Ceratina smaragdula (Image 2e), Xylocopa
latipes (Image 2c), Nomia
sp. (Image 2f) and Lasioglossum sp. (Image
2g). The wasps were Rhynchium
sp. (Image 2h) and Ammophila sp.(Image 2i). The flies were Sarcophaga
sp. (Image 2j) and Eristalinus sp.
(Image 2k). The butterflies included Eurema hecabe
(Image 3a), Leptosia nina
(Image 3b) (Pieridae), Zizula
hylax (Image 3c), Pseudozizeeria
maha (Image 3d), Zizeeria
karsandra, Everes lacturnus (Image 3e) (Lycaenidae)
and Pelopidas mathias (Image
3f) (Hesperiidae) (Table 2). Of these, all bees except Xylocopa
sp. foraged for both pollen and nectar while Xylocopa
sp. and all other foragers foraged for nectar only. All these insects approached the flowers in
upright position, landed on the flat-topped capitulum
and then probed ray and disc florets for nectar. They foraged several florets in a single
visit and made multiple visits to several capitula on
the same plant in quest of forage. They
made frequent visits to capitula of different closely
and distantly spaced plants to collect forage.
Such a foraging behavior was considered to be
promoting both self- and cross-pollination.
The foraging activity pattern of insects showed a definite pattern with
reference to foraging schedule. They foraged
flowers during 08:00-16:00/17:00 hr with peak
foraging during 10:00-12:00 hr (bees, wasps and
flies) and during 10:00–11:00 hr (butterflies)
coinciding well with the standing crop of nectar by that time (Figs. 1,2). Bees made 42%, wasps 13%, flies 11% and
butterflies 34% of total foraging visits (Fig. 3). The body washings of insects collected from
the flowers during peak foraging period revealed that all insects carry pollen
but bees carry the highest number of pollen grains. Further, the mean number of pollen grains
varied with each insect species (Table 3).
Fruiting ecology and seed dispersal
The
fertilized disc florets produce single-seeded cypselas
within two weeks. Natural cypsela set is
86% in ray florets and 98% in disc florets (Table 4). In ray florets, the cypsela is 3–5 mm long,
2.5–3.1 mm wide, pale brown, oval, tangentially flattened with
upwardly-pointing teeth along the marginal wing and 2 short terminal awns
(Image 3g). In disc florets, the cypsela
is 4–5 mm long, 1.8–2.5 mm wide, cylindrical, thickened, 4-sided and
tangentially compressed with 2 or rarely 3 terminal stiff divaricate awns
(Image 3h). The cypselas
of disc florets detach earlier than those produced from ray florets. The stiff echinate
structures of cypselas of both types of florets
enable them to stick readily to hair, fur, clothing and animal skin for
dispersal; the cypselas of disc florets being light
in weight disperse easily to long distances than those of ray florets which are
comparably heavy. Wind dispersed the cypselas efficiently when ambient air is dry. Water also acted as an efficient dispersal
agent for the dispersal of cypselas during the rainy
season. Therefore, zoochory,
anthropochory, anemochory
and ombrohydrochory are the functional forms of seed
dispersal in this plant.
The cypselas of both ray and disc florets are viable and
germinate within a week after their dispersal from the parental plants if the
soil is damp and fertile (Image 3i). Cypselas of both floret types sowed in different pots
filled with fertile soil produced seedlings within a week; germination rate is
60% in ray floret cypselas and 80% in disc floret cypselas. The
seedlings have epigeal germination. The hypocotyl is 10–18 mm long, purplish and
hairless. The cotyledons are elliptic, 5–9 mm long, purplish and shortly
stalked. The paired juvenile leaves are
similar to adult leaves but smaller.
After two weeks, they were transplanted to natural soil and followed for
their life cycle, which is nearly four months.
The observations indicated that both types of cypselas
produce identical individuals and produce both types of cypselas.
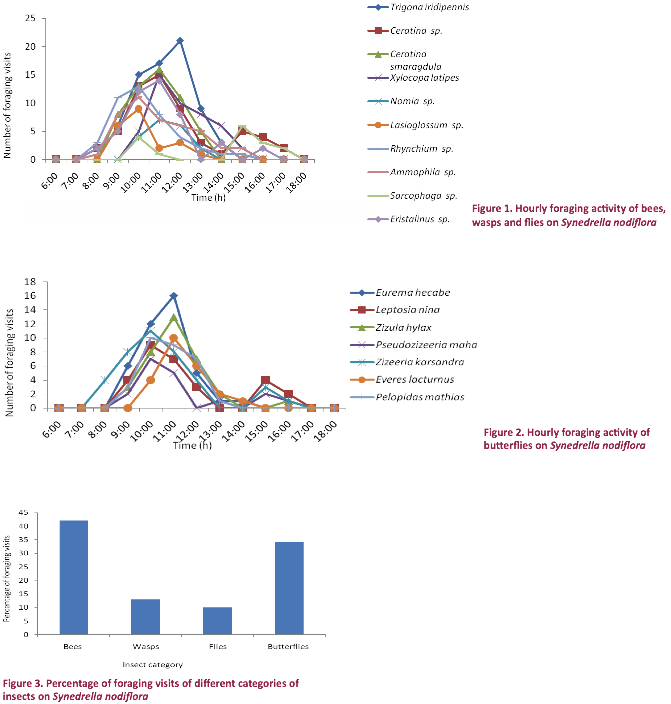
Table 3. Pollen recorded in the body
washings of insect foragers on Synedrella nodiflora
|
Insect species |
Sample size (N) |
Number of pollen grains |
||
|
Range |
Mean |
S.D |
||
|
Trigona iridipennis |
10 |
56-134 |
106.7 |
21.08 |
|
Ceratina sp. |
10 |
25-56 |
37.6 |
8.54 |
|
Ceratina smaragdula |
10 |
21-63 |
48.5 |
11.78 |
|
Xylocopa latipes |
10 |
17-50 |
29.8 |
9.47 |
|
Nomia sp. |
10 |
20-53 |
35.7 |
10.13 |
|
Lasioglossum sp. |
10 |
15-49 |
34.4 |
8.79 |
|
Rhynchium sp. |
10 |
11-42 |
28.2 |
8.31 |
|
Ammophila sp. |
10 |
8-25 |
15.9 |
5.27 |
|
Sarcophaga sp. |
10 |
10-31 |
20.5 |
6.00 |
|
Eristalinus sp. |
10 |
11-26 |
18.4 |
4.08 |
|
Eurema hecabe |
10 |
21-54 |
37.9 |
8.64 |
|
Leptosia nina |
10 |
8-27 |
20.2 |
5.61 |
|
Zizula hylax |
10 |
5 -24 |
21.7 |
6.86 |
|
Pseudozizeeria maha |
10 |
11-28 |
21.5 |
4.60 |
|
Zizeeria karsandra |
10 |
9-36 |
26.4 |
7.47 |
|
Everes lacturnus |
10 |
10-32 |
23.2 |
5.97 |
|
Pelopidas mathias |
10 |
7-24 |
15.7 |
4.37 |
Table 4. Natural cypsela set in Synedrella nodiflora
|
Ray florets |
Disc florets |
||||
|
No. of florets
sampled |
No. of florets
set cypsela |
Cypsela set (%) |
No. of florets
sampled |
No. of florets
set cypsela |
Cypsela set (%) |
|
98* |
84 |
86 |
258** |
253 |
98 |
|
*Sample size: 14; **Sample size:
20 |
|||||

DISCUSSION
Synedrella nodiflora is a herbaceous
weed and grows in soils with sufficient moisture for its rapid germination,
growth, flowering and seed set. It grows
in a very wide range of habitats and tolerates most forms of cultivation
because of its short life cycle (Holm et al. 1997). As an exotic species, it is widely
distributed in both natural habitats and cultivated fields in India (Ansari et
al. 2016). It is one of the widespread
weeds endangering the native flora (Singh et al. 2002) and causing a menace in
cultivated fields due to its invasiveness (Singh et al. 2010). Despite this situation created by this weed,
there are no studies on this species as to how it is able to grow and invade
varied habitats. The present study shows
that S. nodiflora is a therophyte
and produces new sets of populations continuously in damp soils of forest and
agricultural systems. As a result, it
displays vegetative, flowering and fruiting phases simultaneously in different
habitats throughout the year. But, the
rainy season is the best period for its prolific growth in any habitat because
the soil is damp and contains nutrients due to decomposition of organic matter
at this time. Peak flowering occurs
during October-November in different populations. Therefore, it can be said that soil moisture
and nutrient content greatly influence the growth and population size of this
weed in different habitats.
Jeffrey
(2009) stated that Asteraceae members produce capitula consisting of peripherally located ray florets and
centrally located disc florets. The ray
florets are highly specialized in pollinator attraction while disc florets
assume the reproductive function and hence improve the chances for reproductive
success and facilitate a more flexible basis for breeding system evolution than
does a single flower. Further, this
author stated that a particularly diverse trait in the capitulum
is the perianth symmetry exhibited by peripheral ray
and central disc florets; the ray florets display zygomorphic symmetry while
disc florets display actinomorphic symmetry.
The present study shows that S. nodiflora
uses nectar-less ray florets for pollinator attraction and reproductive
function, and nectariferous disc florets exclusively
for reproductive function. The position
of several florets in a capitulum as a unit enables
the plant to maximize reproductive success.
Torices et al. (2011) stated that the
expression of floral sexuality is associated with changes in symmetry, which
has important consequences for the evolution of reproductive biology in Asteraceae. This
generalization is absolutely true in S. nodiflora
because the peripheral zygomorphic ray florets are female while the
actinomorphic disc florets are bisexual.
Chapman & Abbott (2009) reported that the presence of peripheral ray
florets shows a marked effect on attractiveness to pollinators,
cross-pollination rate and fitness for the plant. In S. nodiflora,
the highest fruit set recorded in both ray and disc florets indicate that ray florets
attract pollinator fauna and enhance both self and cross-pollination rate in
both types of florets enabling the plant to increase fitness as an invasive
weed.
Allen
et al. (2011) reported that dichogamous protandry and self-incompatibility are functional in Asteraceae; self-incompatibility occurs at the stigmatic
surface either by failure of germination of self-pollen grains or by the arrest
of pollen tube growth. Harder et al.
(2000) noted that dichogamous protandry
reduces rates of self-fertilization and enhances out-crossing. Nettancourt (2001)
stated that self-incompatibility is another evolutionary strategy to avoid
self-fertilization and inbreeding. The
present study shows that S. nodiflora is dichogamous, protandrous,
self-compatible and self-pollinating.
But, disc florets have a mechanism to prevent spontaneous
self-pollination but not vector-mediated self-pollination. In disc florets, the staminate
phase on day 1 and pistillate phase on day 2
characterize temporal dioecy. These florets are also herkogamous
as they have a physical barrier between the site of pollen deposition and
stigmatic region; herkogamy favors
xenogamy, and pollination occurs during pistillate phase.
The occurrence of self-pollination, however, is possible due to the centripetal
opening of florets in the capitulum on consecutive
days. In this study, fruit set standing
at 86% in ray florets and 98% in disc florets in open-pollinations indicate the
function of self-compatibility and self-pollination in this plant. The pollen/ovule ratio recorded for S. nodiflora (955:1) in this study falls in the range of
pollen/ovule ratio (244.7-2,588) for facultative xenogamy
provided by Cruden (1977) and hence this plant has
facultative xenogamous breeding system with low
self-incompatibility. In this breeding
system, xenogamy enables plants to increase genetic heterogeneity which favors their
establishment in heterogeneous and variable environment (Hsu 2006) while geitonogamy facilitates to increase their population in the
currently growing sites or other sites with similar environment. Therefore, the
widespread and invasive character of S. nodiflora
could be attributable to low self-incompatibility and facultative xenogamy.
Floret
opening in the morning is a common feature among the Asteraceae
(Proctor & Yeo 1978), and it usually occurs before 08:00hr (Mani & Saravanan 1999). In S.
nodiflora also, anthesis
of both ray and disc florets occurs in the morning from 07:00–0900 hr. Within a capitulum,
ray florets open first exposing simultaneously the white ligulate
petal and the receptive stigmatic region by unfolding the stylar
arms to receive pollen from other capitula of the
same or different plants. These florets
cease stigma receptivity by the end of the same day and become non-functional
to receive pollen from the next day onwards.
The disc florets open concentrically on three consecutive days but each
floret shows staminate and pistillate
phases on two consecutive days to avoid spontaneous autogamy and minimize geitonogamy. In Asteraceae, secondary pollen presentation mechanism is an
important characteristic associated with protandry
(Howell et al. 1993) but it is an ancestral feature in this family (Jeffrey
2009). This pollen presentation
mechanism has been considered to be a strategy to improve accuracy in pollen
removal and deposition in order to enhance male and female fitness (Ladd
1994). In the present study, the disc
florets of S. nodiflora display this form of
pollen presentation. In these florets,
the pollen shed from the anthers is brushed by the sterile sweeping hairs
present at the base of the abaxial side of stylar arms when the style branches are joined and inner
stigmatic surfaces are not receptive.
This pollen presentation mechanism is an intermediate mechanism
representative of brush mechanism sensu Leins & Erbar (2006). As the style grows out of the anther tube,
the outer sweeping hairs of the style arms present pollen for pollination. The receptive papillate
stigmatic surface is hidden between the two appressed
style arms, preventing self pollination. During the functionally female phase of the floret which occurs on day 2, the style arms separate
exposing the receptive papillae for the receipt of pollen. The style arms serve as secondary pollen
presenters in the staminate phase and expose
receptive stigmatic surfaces for pollen during pistillate
phase. This type of active pollen
presentation is typical of disc florets of Asteraceae
(Ladd 1994). The secondary pollen
presentation system functional in S. nodiflora
appears to have evolved to enhance the efficiency and accuracy of pollen
exportation and/or pollen reception, thus increasing male and/or female fitness
of the plant (Yeo 1993).
Varatharajan &
Daniel (1984) reported that thrips have an intimate
association with the capitula of Asteraceae. Laughlin (1977) reported that in Asteraceae, the duration of growth and development of thrips synchronizes well with the centripetal disc floral
development whereby thrips, which are mostly pollen
feeders, efficiently use the capitulum for their
growth and survival. Kirk (1997) noted
that thrips puncture the pollen coat and drain the
grains; individual thrips consume more than 800
pollen grains per day. Abrol (2012) noted that thrips
carry pollen ranging from 129–180 pollen grains but the pollen loads carried
vary with the larvae and adults with the latter carrying an increased load due
to greater surface area such as wing fringes, abdominal setae, as well as the
antenna. Grimaldi
& Engel (2005) stated that the higher the pollen production the greater the
level of pollen carrying capacity. Ananthakrishnan (1982) reported that the pollen carrying
capacity could also depend upon the pollen surface and architecture. Pollen grains attached to the body setae, wings
and legs of thrips are dispersed on the stigma by way
of their active movement, rubbing the abdomen on the stigmatic surface,
cleansing of their body parts with their hind legs and also by their
wing-combing mechanism. Kirk (1997) reported
that thrips use the stigma for landing and
take-off. During this process, thrips place the pollen directly on the stigma. The impressive petal colour and corolla tube
of a small flower form is an ideal site to attract thrips
for oviposition, enabling the emerging larvae to
become dusted with pollen in their upward and downward movement eventually
leading to pollination. Ananthakrishnan et al. (1981) reported that heterogamous capitula of Asteraceae facilitate
free movement of both larvae and adults in between the individual florets, and
adults carry a maximum pollen load on the body.
In the present study, Microcephalothrips
abdominalis is the only thrips
species which uses S. nodiflora capitula for its breeding.
The larvae emerge in synchrony with the timing of anthesis
of capitulum which occurs for four consecutive days. The larvae and adults move freely up and down
within and between ray and disc florets in search of pollen and nectar; the
larvae carry less pollen while the adults carry more pollen because of variation
in the surface area of the body.
Further, they use stylar arms for take-off and
landing during which the stigmatic area is dusted with pollen. The feeding activities of larvae and adult thrips within the capitulum
contribute to self-pollination. As there
is a continuous emergence of thrips in synchrony with
sequential anthesis within the capitulum,
the available forage becomes insufficient to meet their food requirement and in
effect they migrate to other capitula on the same
plant or nearby plants in search of forage due to which, chances of
cross-pollination are enhanced. Ananthakrishnan et al. (1981) stated that the thrips living in the heterogamous capitula
of Asteraceae with solitary inflorescences spend more
energy for their visits to other flowers where the food is plenty. In S. nodiflora,
the solitary as well as grouped capitula borne in
leaf axils enable thrips to optimize energy
expenditure to visit and acquire more energy from the forage they collect from
different capitula within the plant. Such an
interaction between S. nodiflora and M. abdominalis benefits both
partners, the former in pollination and the latter in breeding and feeding.
In S.
nodiflora, the peripheral nectar-less yellow ray
florets attract foragers. In ray
florets, the the stylar
arms with exposed receptive stigmatic region standing above the tubular portion
of the corolla is an adaptation for easy pollen deposition and pollination by
the foragers which visit the disc florets for pollen and/or nectar. In the central yellow disc florets, the
narrow tubular corolla containing nectar, pollen accessibility to foragers
within and outside the corolla tube, and pollen characteristics such as
spheroidal shape, tri-colporate apertures and echinate exine are adaptations
for insect-pollination. Wodehouse (1935)
stated that the echinate trait of the pollen grains
is a special adaptation for adherence to insect vectors. Therefore, the characteristics of both ray
and disc florets suggest that this plant is adapted for insect-pollination.
Faegri & Pijl (1979) described the floral characters of
butterfly-pollination. They usually
possess large, white, pink, red, yellow or blue, narrow, tubular flowers with
deep nectaries and nectar guides. Baker & Baker (1982; 1983) categorized two
categories of flowers with reference to flower-butterfly relationships. The first category is “true butterfly
flowers” which are characterized by deep, narrow corolla tubes with relatively
copious sucrose-rich nectar. The second
category is “bee and butterfly flowers” which are characterized by short-tubed
corolla with hexose-rich nectar. In S.
nodiflora, the nectar of disc florets is
sucrose-rich with moderate sugar concentration and sugar content, and has some
essential and non-essential amino acids.
These characteristics of disc florets conform to “true butterfly
pollination syndrome”. The tubular
corolla of ray florets lacking nectar and stamens and having functional pistil
indicates that it is adapted for butterfly-pollination by deception.
In the
present study, butterflies are the consistent and regular foragers throughout
the year for S. nodiflora. The capitula
attract butterflies belonging to pieridae, lycaenidae and hesperiidae but lycaenids are the principal pollinators. It is interesting to note that papilionid butterflies never visited the capitula. The small,
narrow tubate ray and disc florets aggregated into capitula are appropriate for pollination by butterflies which efficiently handle several florets to
collect nectar successfully in a single visit.
In such an act, their proboscis gains contact with the dehisced anthers
situated inside the corolla tube and also with the pollen adhered to sweeping
hairs during staminate phase in disc florets. The proboscis is the carrier of pollen and
the number of pollen grains carried by them vary with
the length of proboscis and the time of nectar collection. In pistillate phase
of disc florets, the butterflies with their proboscis contact the exposed
stigmatic regions and in effect transfer and deposit pollen at this region
effecting pollination. Further, the
butterflies with pollen-laden proboscis pollinate the ray florets while probing
their tubular corolla lacking nectar. In
both disc and ray florets, self-pollination occurs due to simultaneous
occurrence of staminate and pistillate
phases in different disc florets, and only pistillate
phase in ray florets at capitula and plant
level. The standing crop of nectar at
plant or population level is commensurate with the requirement of the butterflies
as sufficient volume of nectar is available at capitulum
level. The butterflies frequent the capitula of the same and/or different closely or distantly
spaced individuals and promote cross-pollination. Their foraging schedule and the peak foraging
activity period coincide well with the availability levels of standing crop of
nectar. The consistent foraging activity
of butterflies on this plant suggests that they use this plant as an important
nectar source. Therefore, S. nodiflora is
principally psychophilous.
In the
present study, it is found that bees, wasps and flies also use the disc florets
of S. nodiflora as pollen and/or nectar
sources, especially during peak flowering season. The foraging behavior
of the bees indicated that they are successful mostly as pollen collectors due
to a slight mismatch between the length of their tongue and the length of the
corolla tube to collect the deeply seated nectar. In case of wasps and flies, their proboscis
length easily facilitates nectar collection from the disc florets. All these insects also probe the ray florets
for nectar without any discrimination although there is no nectar; their visits
to ray florets result in pollination.
All these insects carry pollen on their body, transfer and effect self-
and cross-pollination. Since they show
foraging activity mainly during the peak flowering period, they serve as
supplementary pollinators. Nectar
feeding by the larvae and adults of M. abdominalis
leads to reduced levels of nectar and make all nectar feeding insect species to
increase foraging activity across populations to meet their nectar requirement
and in effect, both self- and cross-pollination rates are enhanced.
Different
authors reported that Synedrella nodiflora produces two types of fruits (Banerji & Pal 1959; Kissmann
& Groth 1992; Rocha 1996). The present study also found that S. nodiflora produces two types of cypselas
within a capitulum.
The cypselas produced from ray florets are
heavier, elliptical, membranous with upwardly-pointing
teeth along the margins and two short terminal awns while those from disc
florets are lighter, cylindrical and tangentially compressed with 2 or 3 stiff
terminal divaricate awns. Chauhan & Johnson (2009) stated that the heavier cypselas produced from ray florets could be related to the
inclusion of additional carbohydrate resources by the plant to help in the
plant emergence while Brandel (2007) attributed the
presence of superior mass in the cypselas produced
from ray florets to the pericarp wing which is developed as an alternative
structure for dispersion. The present
study shows that the differences in morphological features of these two types
of cypselas favour spatial dispersion having
different strategies which promote spreading in
various sites. In a capitulum,
the cypselas of disc florets detach first and
disperse to farther sites while those of ray florets disperse later in the
vicinity of mother plants. The
morphological features such as marginal and terminal teeth in ray floret cypselas and terminal teeth in disc floret cypselas enable them to be dispersed by animals and
humans. Further, wind during dry season
and water during rainy season also disperse them farther away. Therefore, the morphological features of cypselas of S. nodiflora
are adapted for dispersal by multiple vectors, the situation of which is
referred to as “polychory” involving zoochory, anthropochory, anemochory and ombrohydrochory.
The
present study shows that the cypselas of both ray and
disc florets of S. nodiflora are viable and
germinate within a week after their dispersal from the mother plants and
produce new plants. This situation indicates that both types of cypselas do not have dormancy. Similarly, Bradford (1990) reported that both
cypsela types in this plant do not have any difference in the fruit coat
permeability and hence germination responses between them are almost the same;
both cypselas types germinate immediately after
dispersal. On the contrary, Brandel (2004) reported some differences in germination rates
of the two cypsela types in S. nodiflora
and related these differences to the ability of cypselas
to environment perception. Turner (1994)
also reported that fresh cypselas of both types of
florets germinate quickly in a wide range of conditions. The buried cypselas
to 10cm depth in soil remain viable for one year. Chauhan &
Johnson (2009) reported that in S. nodiflora,
the cypselas germinate immediately but ray floret cypselas if buried in soil would remain dormant for several
months and germinate when favourable conditions exist. The present study indicates that heterocarpy in S. nodiflora
is a mixed strategy in which disc floret cypselas
successfully germinate under different conditions in time and space and ray
floret cypselas germinate in restricted environment,
usually near mother plants or the same habitats. The ability of disc floret cypselas offers the chance to colonize new sites, free from
sibling competition or other local sources of stress, whereas the remainder of
the offspring stays in the same habitat (Imbert &
Ronce 2001).
Interestingly, both types of cypselas produce
identical plants and also produce bimorphic cypselas within the capitulum
during flowering period suggesting that they are genotypically
identical but different only by morphology which is
configured by environmental situations.
The study suggests that S. nodiflora
with bimorphic cypselas and
the associated traits is able to grow as a widespread weed in different
habitats and expand its distribution range.
This plant is considered to be a menace in agro-ecosystems. But it is an important food source for bees,
wasps, flies and butterflies. Therefore,
it may be an important component of agro-ecosystems because it plays a
significant role in sustaining communities of insects that contribute to crop
growth. Nevertheless, the information
detailed here on the reproductive system of this weed is important to plan a
management control schedule.
REFERENCES
Abrol, D.P. (2012). Pollination Biology: Biodiversity
Conservation and Agricultural Production. Spring Science+Business
Media, New York, 791pp.
Allen, A.M., C.J. Thorogood,
M.J. Hegarty, C. Lexer
& S.J. Hiscock (2011). Pollen-pistil interactions and
self-incompatibility in the Asteraceae: new insights
from studies of Senecio squalidus
(Oxford Ragwort). Annals of Botany 108: 687–698; https://doi.org/10.1093/aob/mcr147
Ananthakrishnan, T.N. (1982). Thrips and pollination biology. Current Science
51: 168–172.
Ananthakrishnan, T.N. (1993). Bionomics of
thrips. Annual Review of Entomology 38:
71–92; https://doi.org/10.1146/annurev.en.38.010193.000443
Ananthakrishnan, T.N., R. Varatharajan
& K. Gopinathan (1981). Seasonal
periodicity of thrips infesting some Compositae in relation to pollination. Proceedings of Indian National Science
Academy B47: 811–815.
Ansari, A.A., S.S. Gill, Z.K. Abbas &
M. Naeem (2016). Plant Biodiversity: Monitoring, Assessment
and Conservation. CABI, New Delhi, 628pp.
Baker, H.G. & I. Baker (1982). Chemical constituents of nectar in
relation to pollination mechanisms and phylogeny, pp. 220–230. In: Nitecki,
M.H. (ed.). Biochemical Aspects of Evolutionary Biology.
The University of Chicago Press, Chicago.
Baker, H.G. & I. Baker (1983). A brief historical review of the chemistry
of floral nectar, pp. 126–152. In: Bentley, B. & T. Elias (eds.). The Biology
of Nectaries. Columbia University Press,
New York.
Banerji, I. &
S. Pal (1959). A contribution to the life history of Synedrella
nodiflora Gaertn. The Journal of Linnean Society Botany 55: 810–817.
Bhatti, J.S. (1980). Species of the genus Thrips from India (Thysanoptera).
Systematic Entomology 5: 109–166.
Bradford, K.J. (1990). A water relations
analysis of seed germination rates. Plant Physiology 94: 840–849; https://doi.org/10.1104/pp.94.2.840
Brandel, M. (2004). Dormancy and germination of heteromorphic achenes of Bidens frondosa. Flora 199:
228–233.
Brandel, M. (2007). Ecology of achene dimorphism in Leontodonsaxatilis. Annals of Botany 100: 1189–1197; https://doi.org/10.1093/aob/mcm214
Burkill, H.M. (1985). The Useful
Plants of West Tropical Africa. Royal Botanical Gardens, Kew, 981pp.
Chapman, M.A. & R.J. Abbott (2009). Introgression of fitness
genes across a ploidy barrier. New Phytologist
186: 63–71; https://doi.org/10.1111/j.1469-8137.2009.03091.x
Chauhan, B.S. &
D.E. Johnson (2009). Seed germination and seedling emergence of Synedrella
(Synedrella nodiflora)
in a tropical environment. Weed Science 57: 36–42.
Cruden, R.W. (1977). Pollen-ovule ratios: a conservative
indicator of breeding systems in flowering plants. Evolution 31: 3246; https://doi.org/10.1614/WS-08-015.1
Dafni, A., P.G. Kevan
& B.C. Husband (2005). Practical Pollination Biology. Enviroquest Ltd., Cambridge, 583pp.
Davidse, G., M. Sousa-Pena, S. Knapp & F.C.
Cabrera (2015). Asteraceae. In: Davidse,
G., M.S. Sanchez, S. Knapp & F.C. Cabrera (eds.). Flora Mesoamericana. Universidad NacionalAutonoma
de Mexico, Mexico.
Faegri, K. &
L. Pijl (1979). The Principles of
Pollination Ecology. Pergamon Press, New York, 244pp.
Grimaldi, D. &
M.S. Engel (2005). Evolution of the Insects. Cambridge University Press, Cambridge, UK, 755pp.
Gunathilagaraj, K., T.N.A. Perumal,
K. Jayaram & M.G. Kumar (1998). Field Guide: Some South Indian Butterflies. Niligiri Wildlife and Environmental Association, Niligiri,
274pp.
Harder, L.D., S.C.H. Barrett & W.W.
Cole (2000). The mating consequences of sexual segregation within inflorescences of
flowering plants. Proceedings of the Royal Society of London B: Biological Sciences 267:
315–320.
Holm, L.G., J. Doll, E. Holm, J.V. Pancho & J.P. Herberger
(1997). World
Weeds: Natural Histories and Distribution. John Wiley & Sons
Inc., New York, 1129pp.
Howell, G.J., A.T. Slater & R.B. Knox
(1993). Secondary pollen presentation in angiosperms and its biological
significance. Australian
Journal of Botany 41: 417-438.
Hsu, H.M. (2006). Implication
of the invasiveness of Bidens pilosa
var. radiata Sch. Bip. by studying its
superiority over Bidens bipinnata
L. MS Thesis, National Taiwan University, Taipei, 193pp.
Idu, M. &
H.I. Onyibe (2007). Medicinal plants of Edo State, Nigeria. Research Journal of Medicinal Plant
2: 32–41.
Imbert, F. &
O. Ronce (2001). Phenotype plasticity for dispersal ability
in the seed heteromorphic Crepis sancta
(Asteraceae). Oikos
93: 126–134; https://doi.org/10.1034/j.1600-0706.2001.930114.x
Jeffrey, C. (2009). Evolution of
Compositae flowers, pp. 131–138. In: Funk, V., A. Susanna, T.F. Stuessy & R.J. Bayer (eds.). Systematics,
Evolution, and Biogeography of Compositae.
International Association for Plant Taxonomy, Vienna.
Kirk, W.D.J. (1997). Feeding. pp.
65–118. In: Lewis, T. (ed.). Thrips as Crops Pests. CAB International, Oxon, UK.
Kissman, K.G. &
D. Groth (1992). Plantasinfestantes e nocivas. BAST, Sau Paulo, 349–351pp.
Kunte, K. (2007). India - A Lifescape:
Butterflies of Peninsular India. Universities Press, Hyderabad, 254pp.
Ladd, P.G. (1994). Pollen presenters in the flowering plants
- form and function. Botanical Journal of Linnean
Society 115: 165–195; https://doi.org/10.1111/j.1095-8339
Laughlin, R. (1977). The gum tree
thrips, Isoneurothrips
australis Bagnall,
Survival at different temperature and humidities and
its relation to capacity for survival. Australian Journal of
Ecology 2: 391–398.
Leins, P. &
C. Erbar (2006). Secondary pollen presentation syndromes of
the Asterales - a phylogenetic perspective. Botanische
Jahrbucher fur Systematik
127: 83–103.
Mani, M.S. & J.M. Saravanan
(1999). Pollination ecology and evolution in Compositae
(Asteraceae). Science Publishers, New Hampshire, 166pp.
Nettancourt, D. (2001). Incompatibility and Incongruity in Wild
and Cultivated Plants. Springer-Verlag, Berlin, Germany, 322pp.
Proctor, M.C.F. & P. Yeo (1978). The Pollination of Flowers. William Collins Sons
& Co. Ltd., Glasgow, 418pp.
Rocha, O.J. (1996). The effects of a achene heteromorphism on the dispersal capacity of Bidenspilosa L. International Journal of Plant
Sciences 157: 316–322; https://doi.org/10.1086/297351
Singh, M.P., B.S. Singh & S. Dey (2002). Plant Biodiversity and Taxonomy. Daya Publishing House, Delhi, 260pp.
Singh, K.P., G. Dhakre
& V. Daboriya (2010). Phenology
and reproductive biology of Parthenium hysterophorus L. Advances in Pollen-Spore Research
28: 143–148.
Torices, R., M.
Mendez & J.M. Gomez (2011). Where do monomorphic sexual systems fit in
the evolution of dioecy? Insights
from the largest family of angiosperms. New Phytologist
190: 238–248; https://doi.org/10.1111/j.1469-8137.2010.03609.x
Turner, B.L. (1994). Taxonomic
study of the genus Synedrella (Asteraceae, Heliantheae). Phytologia 76: 39–51.
Varatharajan, R. &
M. Daniel (1984). Studies on soil pupation in some phytophagousthrips. Journal of Soil
Biology and Ecology 4: 116–123.
Wodehouse, R.P. (1935). Pollen Grains: Their Structure,
Identification, and Significance in Science and Medicine. McGraw-Hill,
New York, 573pp.
Yeo, P.F. (1993). Secondary pollen presentation: form,
function and evolution. Plant Systematics and Evolution (Supplement) 6:
1–268.