Journal of
Threatened Taxa | www.threatenedtaxa.org | 26 September 2018 | 10(10):
12363–12367
Delayed peracute capture myopathy in a Himalayan Ibex Capra sibirica
(Mammalia: Cetartiodactyla: Bovidae)
Umar Nazir Zahid 1, Latief
Mohammad Dar 2, Umar Amin 3, Showkat
Ahmad Shah 4, Rashid Yahya Naqash 5, Dil Mohammed
Makhdoomi 6, Shayuaib
Ahmad Kamil 7 & Intesar Suhail 8
1,5,8 Department of Wildlife Protection,
Kashmir, Jammu & Kashmir 190005, India
2 Department of Animal Husbandry, Kashmir, Jammu & Kashmir 190001, India
3,4,7 Division of Veterinary Pathology, 6
Division of Veterinary Surgery and Radiology, Shere-e-Kashmir
University of Agriculture Sciences and Technology, Kashmir, Jammu & Kashmir
191121, India
1drumarnazir@gmail.com (corresponding
author), 2shaheenlatief@gmail.com, 3umaramin46@gmail.com,
4vetshowkat@gmail.com, 5hangulnaqash@yahoo.com, 6dmmakhdoomi@gmail.com,
7kamilshoaib@gmail.com, 8intesar.suhail@gmail.com
doi: https://doi.org/10.11609/jott.3899.10.10.12363–12367
Editor: Ulrike Streicher, Cascades
Raptor Center, Eugene, USA. Date of publication: 26 September 2018 (online &
print)
Manuscript details: Ms # 3899 |
Received 17 November 2017 | Final received 22 May 2018 | Finally accepted 25
August 2018
Citation: Zahid, U.N., L.M. Dar, U. Amin, S.A.
Shah, R.Y. Naqash, D.M. Makhdoomi,
S.A. Kamil & I. Suhail (2018). Delayed peracute capture
myopathy in a Himalayan Ibex Capra sibirica
(Mammalia: Cetartiodactyla: Bovidae). Journal of Threatened
Taxa 10(10): 12363–12367; https://doi.org/10.11609/jott.3899.10.10.12363-12367
Copyright: © Zahid et al. 2018. Creative Commons Attribution 4.0
International License. JoTT allows
unrestricted use of this article in any medium, reproduction and distribution
by providing adequate credit to the authors and the source of publication.
Funding: None.
Competing interests: The authors declare no competing interests.
Abstract: The present study documents a unique
case of capture myopathy as a fatal consequence of the capture and rescue of a
Himalayan Ibex kid. The ibex died 48
hours after capture without any visible clinical signs. Necropsy revealed alterations in kidneys with
necrosis of the renal cortex, degeneration of tubular cells and congestion as
the main histopathological alterations. Lesions in the heart consisted of multifocal
degeneration of myofibres as well as hyalinization
and nuclear degeneration with pyknosis. Skeletal muscles appeared macroscopically
normal but on histopathology showed mild to moderate degeneration and
fragmentation with intermittent loss of striation. The pathological findings were indicative of peracute capture myopathy.
To our knowledge this is the first report of capture myopathy in a
Himalayan Ibex from India underlining the importance of understanding the
causes of mortality in such wild species as a prerequisite to their successful
conservation.
Keywords: Capture, conservation, myopathy, Ibex,
necropsy, pathological findings, rescue.
The
Himalayan Ibex Capra sibirica is a member of
the family Bovidae, sub-family Caprinae
and is a true goat species. Being a
‘sturdy, thick-set goat’ (Prater 1980), the animals
have a short broad face with a long beard in males but short one in females
(Schaller 1977). The geographic distribution
of Himalayan Ibex includes parts of China (Reading & Shank 2008; Xu et al. 2012), India (Gaston et al. 1983; Fox & Johnsingh 1997; Namgail 2006),
Afghanistan (Heptner et al. 1961; Habibi
1997), north-eastern Uzbekistan, Tajikistan, Kyrgyzstan, Pakistan (Reading
& Shank 2008; Li et al. 2015) and the Karakoram, the Himalaya and the
Trans-Himalayan regions of Jammu & Kashmir (Fox & Johnsingh
1997). In India, the Himalayan Ibex is
protected and included in the Schedule I of the Indian Wildlife Protection Act
1972 (Anon 1992). And its conservation
is a priority.
Conservation
plans are developed for wild animals around the world, in which capture and
rescue operations are of paramount importance.
Attempted in the interest of conservation of the concerned wild animals,
capture can be detrimental causing extreme stress and fear in rescued wild
animals leading to capture myopathy and eventual death (Spraker
1982; Ebedes & Raath
1999; McLaren et al. 2007).
Capture
myopathy or exertion rhabdomyolysis is a metabolic
muscle disease of wild mammals and birds (free ranging and captive) associated
with the stress of capture, restraint and transportation (Williams & Thorne
1996). The four clinical syndromes of
capture myopathy documented in wild animals include capture shock syndrome,
ataxic myoglobinuric syndrome, delayed peracute syndrome and ruptured muscle syndrome (Spraker 1993). The
delayed peracute syndrome is usually seen in animals
in captivity for at least 24 hours.
These animals appear normal while undisturbed, but if recaptured or
suddenly stressed they die within several minutes. The pathogenesis of the syndrome is a complex
phenomenon confronting wildlife experts since ages, however, it is argued that
the increase in stress-related catecholamines affects
both cardiac and muscular systems, causing severe muscle damage, rhabdomyolysis and myoglobinuria
(Spraker 1993).
All the stages of the syndrome are potentially fatal due to cardiogenic
shock, renal failure, metabolic disorders or chronic cardiac damage (Spraker 1993).
Materials and
Methods
On 23
August 2017, an orphan Himalayan Ibex kid was rescued from Mahaguns
Top Pahalgam (35.1720N & 75.5010E). The female Ibex kid was found abandoned by
the field staff of the department who later captured the animal by physical
handling apparently without use of any anaesthetic, tranquilizer or sedative.
The kid was translocated to the mini zoo of Pahalgam (34.0310N & 75.3090E) in a transport
box and was then placed in an observation and quarantine room. The observation room is situated away from
the main enclosures with very little human interference, and is maintained in a
way to simulate a natural environment for the rescued animals. Strict hygiene is maintained with foot baths containing bactericidal and virucidal
agents, which are placed both at the entry and exit points. Separate utensils, equipments,
clothing are
provided for the personnel attending the animals in the quarantine area. Immediately after being rescued the animal
was examined; it was approximately three months old, weighed 4.4kg and was
found in good body condition showing no apparent signs of capture
myopathy. The animal was further
monitored closely for any signs of capture myopathy or abnormal behaviour. The animal died 48 hours after being captured
without showing specific clinical signs.
A complete necropsy was conducted within two hours of death. Samples were taken from the heart, kidneys,
liver, lungs and skeletal muscles and were fixed in 10% neutral buffered
formalin and embedded in paraffin wax, and sections (4mm) were stained with hematoxylin and eosin for routine histopathological
examination.
Results
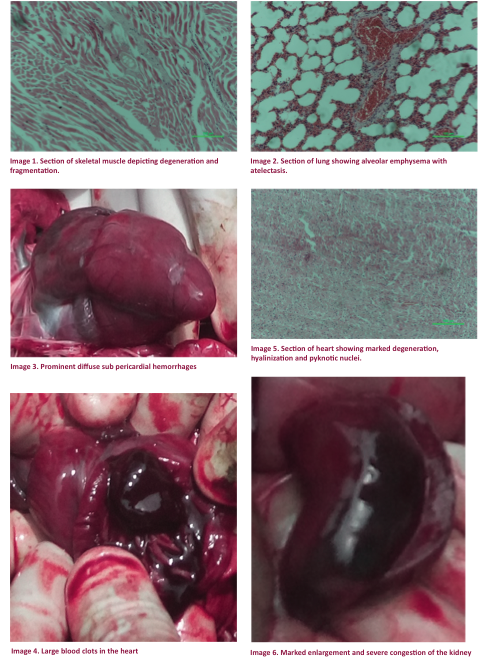
The
external examination of the carcass revealed no lesion or violence inflicted
injury. Also on dissection most organs
appeared grossly normal only the heart showed focal to diffuse sub pericardial haemorrhages with
clotted blood in all the four chambers (Images 3 & 4). But histopathological
examination revealed changes in kidneys, heart, liver and skeletal
muscles. The skeletal muscles showed
mild to moderate degeneration and fragmentation with intermittent loss of
striation (Image 1). Vascular congestion
was found in the liver. The lung parenchyma
showed focal alveolar emphysema with atelectasis as a main histopathological
feature (Image 2). Myocardial lesions
consisted of multifocal degenerative changes of myofibres,
hyalinization and nuclear degeneration with pyknosis
(Image 5). Both the kidneys were found
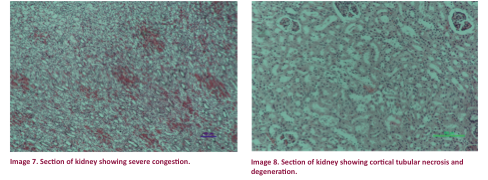
to have developed hydronephrosis with severe
congestion (Image 6). Renal cortical
necrosis, degeneration of tubular cells and congestion were the marked the
changes in the renal parenchyma (Image 7 & 8). Some other changes were increased bowman’s
space with or without serous exudate.
The histopathological changes in different
organs were suggestive of peracute capture myopathy.
Discussion
Capture
myopathy is likely to occur when the capture procedure is tedious involving
vigorous exercise, scaring and tense situations or the excessive use of
tranquilizers. In this case the subject
animal developed capture myopathy in absence of all these factors and the
myopathy was in this case only caused by stress. Assessment of stress would have required
measuring of cortisol levels in the animal after capture so that treatment
measures could have been initiated. The
gross changes observed during post-mortem examination in heart and kidneys
indicated that the animal collapsed due to acute cardiac and renal failure both
of which are the manifestations of rhabdomyolysis (Spraker 1993; Guis et al. 2005; Herráez et al. 2007).
Renal changes leading to nephrosis and multiorgan failure as observed in this case have been
previously reported to be the central pathway of capture
myopathy (Montane et al. 2002; Herráez
et al. 2007; Nuvoli et al. 2014). The myocardial lesions are attributed to
elevated concentrations of endogenous catecholamines
during stress and trauma (Jiang & Downing 1990; Harrez
et al. 2007). Myocardial lesions are
also frequently implicated as an important reason for sudden death under
extreme stress in wild animals and birds (Turnbull & Cowan 1998). These findings are
supported by Wallace et al. (1987) who reported similar myocardial lesions in
case of acute and delayed capture myopathy in African wild ungulates. The changes in skeletal muscles observed in
the present case can be attributed to exertion, trauma and polysaccharide
storage myopathy during rescue and capture procedure leading to ischemia of
muscles and subsequent myocytolysis (Montane et al. 2002; Guis et al.
2005; Nuvoli et al. 2014). Similar findings in liver
and lungs are reported by McAllum (1978) in a
study of capture myopathy in Red Deer.
The
case has been described as delayed peracute capture
myopathy due to the fact that the ibex kid was apparently normal up to 48 hours
after capture followed by sudden peracute death. Absence of prominent clinical signs and
presence of characteristic histopathological findings
in different organs further supported this diagnosis. The classification of this case as delayed
capture myopathy follows Spraker (1993). And like previous studies, the present study
also supports the fact that wild animals like the Himalayan Ibex capture
myopathy is a fatal consequence of stress during capture and handling. Thus, wildlife personnel should exercise
extreme care during trapping, handling and transportation of such endangered
wild animals.
To the
our knowledge this is the first report of capture myopathy in a Himalayan Ibex
from India warranting further studies of the causes of mortality in such wild
species as a prerequisite for a successful conservation programme. Moreover, special attention needs to be paid
to issues including animal welfare and qualification and skills of the
personnel who manage capture and rescue operations.
References
Anonymous (1992). The Indian Wildlife (Protection) Act,
1972, Government of India, Natraj Publishers,
Dehradun.
Ebedes, H. &
J.P. Raath (1999). Use of tranquilizers in
wild herbivores, pp. 575–585. In: Fowler, M.E. & R.M. Miller (eds.).
Zoo & Wild Animal Medicine. Current therapy 4. WB Saunders Company, Philadelphia,
Pennsylvania.
Fox, J.L. & A.J.T. Johnsingh
(1997). Wild
sheep and goats and their relatives: status survey and conservation action plan
for Caprinae. IUCN, Gland,
Switzerland, 215–231pp.
Gaston, A.J., P.J. Garson & M.L.
Hunter (1983). The status and conservation of forest wildlife in Himachal Pardesh, Western Himalayas. Biological Conservation 27:
291-314.
Guis, S., J.P. Mattei,
P. Cozzone, & D. Bendahan
(2005). Pathophysiology and clinical presentations of rhabdomyolysis.
Joint Bone Spine 72: 382–391.
Habibi, K. (1997). Wild sheep and goats and their relatives:
status survey and conservation action plan for Caprinae.
IUCN, Gland, Switzerland, 204–211pp.
Heptner, V.G., A.A. Nasimovich&
A.G. Bannikov (1961). Mammals of the
Soviet Union. Artiodactyla and Perissodactyla. VysshayaShkola,
Moscow, USSR, 1–776 pp.
Herráez, P., E. Sierra, M. Arbelo,
J.R. Jaber, A.E. de Los Monteros
& A. Fernández (2007). Rhabdomyolysis and myoglobinuric nephrosis
(capture myopathy) in a striped dolphin. Journal of
Wildlife Diseases 43: 770–774.
Jiang, J.P. & S.E. Downing (1990). Catecholamine cardiomyopathy: Review and
analysis of pathogenetic mechanisms. Yale Journal of Biology and Medicine 63: 581–591.
Li, Y., Y.Q. Yu & L. Shi (2015). Foraging and bedding
site selection by Asiatic Ibex (Capra sibirica)
during summer in central Tianshan Mountains. Pakistan
Journal of Zoology 47: 1-6.
McAllum, H.J.F. (1978). Post capture myopathy syndrome in Red
Deer (Cervus elaphus).
MVSc Thesis. Massey University.
McLaren, G., C. Bonaic
& A. Rowan (2007). Animal welfare and conservation: measuring stress in the wild. In:
Macdonald, D & K. Service (eds.). Key Topics in
Conservation Biology. Wiley, Hoboken, New Jersey,
120–133pp.
Montane, J., I.
Marco, X. Manteca, J. Lopez & S. Lavin (2002). Delayed acute capture myopathy in three
roe deer. Journal of Veterinary Medicine. A, Physiology, Pathology, Clinical Medicine 49:
93–98.
Namgail, T. (2006). Winter habitat partitioning between
Asiatic Ibex and Blue Sheep in Ladakh, northern
India. Journal of Mountain Ecology 8: 7-13.
Nuvoli, S., G.P. Burrai, F.N. Secci, G.M. Columbano, L. Careddu & M. Mandas (2014). Capture myopathy in a corsican
Red Deer Cervus elaphus
corsicanus (Ungulata: Cervidae). Italian Journal of Zoology 457–462.
Prater, S.H.
(1980). The Book of Indian
Animals. BNHS, Bombay, 324pp.
Reading, R. & C. Shank (2008). Capra sibirica. The IUCN Red List of Threatened Species
2008: e.T42398A10695735. http://doi.org/10.2305/IUCN.UK.2008.RLTS.T42398A10695735.en.
Downloaded on 19
September 2018.
Schaller, G.B. (1977). Mountain Monarchs: Wild Sheep and
Goats of the Himalaya. Chicago, Chicago University Press,
425pp.
Spraker, T.R. (1982). An overview
of the pathophysiology of capture myopathy and related conditions that
occur at the time of capture of wild animals, pp. 83–117. In: Nielsen, L., J.C. Haigh & M.E. Fowler
(eds.). Chemical immobilization of North American Wildlife.
Wisconsin Humane Society, Milwaukee, Wisconsin,
Spraker, T.R. (1993). Stress and capture myopathy in artiodactylids, pp. 481–488. In: Fowler, M.E. (eds.). Zoo & Wild Animal Medicine, Current Therapy, 3rd Edition.
W.B. Saunders, Philadelphia.
Turnbull, B.S. & D.F. Cowan (1998). Myocardial contraction band necrosis in
stranded cetaceans. Journal of Comparative Pathology 118:
317–327.
Wallace, R.S., M. Bush & R.J. Montali (1987). Deaths from exertional
myopathy at the National Zoological Park from 1975 to 1985. Journal of Wildlife
Diseases 23: 454–462.
Williams, E.S. & E.T. Thorne (1996). Exertional myopathy (capture myopathy), pp. 181-193. In: Fairbrother,
A., L.N. Locke & G.L. Hoff (eds.). Noninfectious Diseases of Wildlife, 2nd Edition. Manson
Publishing/Veterinary Press, London.
Xu, F., M. Ma,
Y. Wu & W. Yang (2012). Winter daytime activity budgets of Asiatic Ibex Capra
sibirica in Tomur
National Nature Reserve of Xinjiang, China. Pakistan
Journal of Zoology 44:389–392.