Journal of Threatened Taxa |
www.threatenedtaxa.org | 26 October 2018 | 10(11): 12577–12581
A new report on the
clasper movements of a captive Sand Tiger Shark Carcharias taurus
(Lamniformes: Odontaspididae) and a possible reason for the behaviour
Helen Colbachini 1, Rafael Caprioli Gutierrez
2 , Cristiane Schilbach Pizzutto 3 & Otto Bismarck Fazzano Gadig 4
1,4 Elasmobranch Research Lab, São Paulo
State University, Coastal Campus of São Vicente, São Vicente, SP 11330-900,
Brazil
2 São Paulo Aquarium, R. Huet Bacelar, 407 - Vila São José, São Paulo, SP 04275-000, Brazil
3 Animal Reproduction Department, São
Paulo University, School of Veterinary Medicine and Zootechnics, São Paulo, SP
05508-270, Brazil
1 hcolbachini@gmail.com (corresponding
author), 2 rafael@aquariodesaopaulo.com.br, 3 cspizzutto@yahoo.com.br, 4 gadig@clp.unesp.br
Abstract: Elasmobranchs present four
clasper movements, which can be seen in different contexts from mating to no
obvious reason. Three movements have
been reported in Carcharias taurus and here the first occurrence of
clasper flaring in this species is described.
Clasper flaring was observed while other species were in a reproductive
state and their aggressive behaviour towards the subject of this study was also
observed.
Keywords: Agonistic behavior, Carcharias
taurus, elasmobranch, Grey Nurse Shark, reproduction, reproductive
behaviour.
doi: https://doi.org/10.11609/jott.3852.10.11.12577-12581
Editor: E. Vivekanandan, Central Marine Fisheries Research
Institute, Chennai, India. Date of publication: 26 October
2018 (online & print)
Manuscript
details: Ms # 3852 | Received 23 October 2017 | Final received 25
August 2018 | Finally accepted 22 September 2018
Citation: Colbachini,
H., R.C. Gutierrez, C.S. Pizzutto & O.B.F. Gadig (2018).
A new
report on the clasper movements of a captive Sand Tiger Shark Carcharias
taurus (Lamniformes: Odontaspididae) and a possible reason for the
behaviour. Journal of
Threatened Taxa 10(11): 12577–12581; https://doi.org/10.11609/jott.3852.10.11.12577-12581
Copyright: ©
Colbachini et al. 2018.
Creative Commons Attribution 4.0 International License. JoTT allows
unrestricted use of this article in any medium, reproduction and distribution
by providing adequate credit to the authors and the source of publication.
Funding: Aquário de São Paulo.
Competing interests: The authors declare no competing interests.
Acknowledgments: We
thank the staff of the Aquário de São Paulo who have maintained this species
and provided all support needed to this world.
We are also grateful to G. Stefanelli, C. Colbachini and R.A. Moreira
for providing helpful comments on the manuscript.
Elasmobranchs
have an external structure used to copulate, known as a clasper. This structure has the ability of movement by
itself in order to facilitate copulation (Gilbert & Heath 1972). Four clasper movements have been described,
as follows: flexion (movement of individual claspers backwards and forwards);
splaying (the male contorting itself and opening his claspers up to 900
to the body axis); crossing (claspers crossed with tips posterolateral); and
flaring (clasper flexed and bent forward with their distal ends spread) (Gordon
1993; Compagno 2001; Ritter & Compagno 2013).
In
lamnoid sharks, the clasper skeleton is constituted of the clasper shaft on the
anterior region and the clasper glans on the posterior part. This second region presents a structure
called a clasper hook, which helps the male to anchor itself to the female
during copulation (Gilbert & Heath 1972; Compagno 2001). Specifically in the Grey Nurse Shark Carcharias
taurus, just the first three clasper movements have been reported (Gordon
1993; Compagno 2001); and it is believed that clasper flexion occurs in
different contexts, from mating to without an obvious reason (Myrberg &
Gruber 1974; Gordon 1993).
There
are two hypotheses for the causation of these movements in elasmobranchs: an
agonistic cause (Martin 2007) and a pre-/post-copulation behaviour related to
sperm transportation (Gilbert & Heath 1972; Ritter & Compagno 2013). Although these theories could explain some of
the reports, they are not applicable to all the circumstances. Therefore, here we report the first
occurrence of clasper flaring in Carcharias taurus and present a new
hypothesis for its causation, which would also explain why these two hypotheses
have been made.
Materials and Methods
The subject of study
Our subject was an adult male Carcharias
taurus that was 1.9m length, 80kg in weight and between 20-23 years
old. This individual was housed in the
Oceanarium tank at Aquário de São Paulo, São Paulo, Brazil, which is a 1,000m³
enclosure with depths varying from 1.4m in the superior area to 5m in the
lateral areas.
Besides this specimen, the tank was
inhabited by eight Nurse Sharks Ginglymostoma cirratum, two Brownbanded
Bamboo Sharks Chiloscyllium punctatum, five Southern Stingrays Hypanus
americanus and 10 bony fishes.
This was an opportunistic data
collection, since the main objective of the research was to better understand
the behavior of the subject. After the
first observation of the clasper movements, a new focus has been given to the
study. So, a focal all-occurrences sampling protocol was used due to the low
frequency of the observed movements (Altmann 1974; Lehner 1996). We recorded the type of clasper movements,
time of occurrence, duration and additional observations (such as other
inhabitants’ reproductive and aggressive behaviors).
Observations were made for 72 hours between
January and April 2015; 36 hours of this total were realized between 09:00
and10:00 hr and the remainder, between 21:00 and 22:00 hr. At night time, the tank’s light was turned
off since the aquarium does not work at this period. So, the observation was made with the aid of
a flashlight. However, considering that
unexpected changes in the lighting intensity can cause stress in elasmobranchs
(Powell et al. 2004), the lights were turned on for 30 minutes before all
nocturnal observations. Since this study
was purely observational, it was not considered necessary to ask for an ethical
permission.
Results
Clasper flexion and splaying were
not performed by the subject in this study.
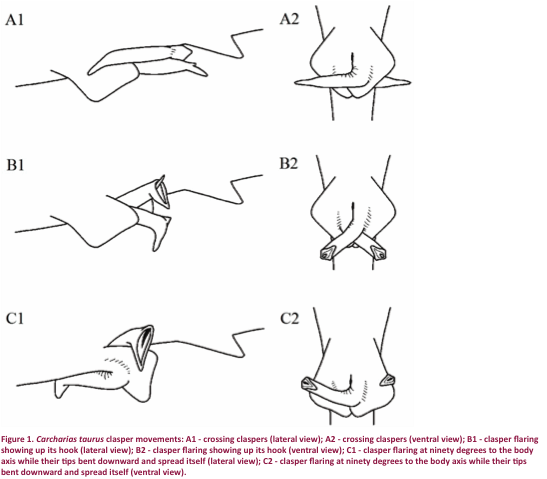
We, however, observed two clasper movements: clasper crossing (CC)
(clasper crossed with tips posterolateral) and clasper flaring (CF). This last movement consists of the clasper
crossed at 90º to the body axis while their tips bent downward and spread
itself, showing up its clasper hook (Fig. 1) (supporting information), that has
never been reported on this species before.
Clasper crossing was observed on
four nights, counting a total of seven occurrences in this work, while clasper
flaring was observed on six days, adding up to seven occurrences (Table
1). These movements were observed while
two other species (Ginglymostoma cirratum and Hypanus americanus)
were at a reproductive stage, and so, some aggressive behaviour of these
inhabitants toward the subject of this study (Table 1) was observed.
Table 1. Description on occurrences of clasper movements in a
captive Carcharias taurus. Additional observations are given on the
context in which the behaviour has been performed.
|
Day |
Hour |
Clasper movements |
Duration (seconds) |
Additional observations |
|
07.i.2015 |
21:44 21:56 21:59 |
CC CC CC |
50 60 20 |
-The subject was contorting itself while doing these behaviours -The nurse sharks of the tank were stalking the subject at 21:53 |
|
09.ii.2015 |
21:21 21:22 |
CF CC |
60 480 |
|
|
05.iii.2015 |
21:04 21:45 |
CF CF |
60 45 |
-Male nurse sharks were active and one of them performed CC at
21:01 |
|
09.iii.2015 |
21:40 |
CF |
30 |
-After the clasper movements, the subject contorted itself for
20 seconds |
|
16.iii.2015 |
21:53 |
CC |
20 |
|
|
14.iv.2015 |
21:33 |
CF |
35 |
|
|
15.iv.2015 |
21:47 21:47 21:48 |
CC CF CC |
10 15 25 |
-Before the clasper movements, the nurse sharks stalked the
subject -One male Southern Stingray performed clasper crossing at 21:11 |
|
24.iv.2015 |
21:53 |
CF |
90 |
-One female Southern Stingray released one egg capsule at 21:17 |
CC - clasper crossing (clasper
crossed with tips posterolateral); CF - clasper flaring (clasper crossed at
ninety degrees to the body axis while their tips bent downward and spread
itself, showing up its clasper hook).
Discussion
Clasper movements have been reported
for various elasmobranch species: Bullhead Shark Heterodontus francisci,
Ginglymostoma cirratum, Carcharias taurus, Gray Reef Shark Carcharhinus
amblyrhynchos, Silky Shark Carcharhinus falciformis, Chain Catshark Scyliorhinus
rotifer, Bonnethead Shark Sphyrna tiburo, Cape Shark Squalus
acanthias, Whitetip Reef Shark Triaenodon obesus, Clearnose Skate Raja
eglanteria, Hypanus americanus, Giant Manta Ray Manta
birostris and Bat Ray Myliobatis californica (Myrberg & Gruber
1974; Klimley 1980; Tricas 1980; Gilbert 1984; Luer & Gilbert 1985;
McKibben & Nelson 1986; Uchida et al. 1990; Gordon 1993; Carrier et al.
1994; Yano et al. 1999; Pratt & Carrier 2001; Chapman et al. 2003; Galván-Tirado
et al. 2014). Most of these movements
correspond to clasper flexion and, in some cases, clasper crossing.
During copulation, some species are
known to expand and spread the tip of the inserted clasper to anchor the male
securely to the female (Gilbert 1984; Luer & Gilbert 1985). Moreover, a Triaenodon obesus and a
Flapnose Ray Rhinoptera javanica performed this behavior for a few
minutes exactly after mating as witnessed by Uchida et al. (1990) in a captive environment. The expanded and spreading tip’s condition
observed on those studies are similar to those found in this work, besides the
fact that our subject did not exhibit the behavior in a reproductive context.
In general, movements of claspers
occur as a pre-copulatory behavior (Myrberg & Gruber 1974; Gordon
1993). Nevertheless, studies on Sphyrna
tiburo and Grey Bamboo Shark Chiloscyllium griseum have reported
these movements outside of mating events without being able to identify a cause
for their occurrences (Myrberg & Gruber 1974; Pratt & Carrier
2001). Martin (2007) has analysed
agonistic behaviors in elasmobranchs and has pointed out that clasper flexion
can be included in this classification despite its uncommon frequency.
During the mating season, agonistic
behaviors are carried out as sexual conflict and may be directed toward
conspecifics and other species which inhabit the same habitat (Gordon 1993;
Martin 2007). In Ginglymostoma
cirratum, aggressive behaviors are required to acquire and mate with
females (Pratt & Carrier 2001) and can also be addressed in interspecific
interactions (Henningsen et al. 2004).
The clasper movements here reported
occurred at night, which agree with the nocturnal activity of the species
(Compagno 2001; Hannon & Crook 2004; Barker et al. 2011). The manifestation of these movements occurred
at the same time of pre-copulatory and aggressive behaviors of Ginglymostoma
cirratum’s and Hypanus americanus’s specimens of the tank.
The Sand Tiger Sharks have been
studied in captivity by Henningsen et al. (2004). These studies have reported that intrasexual
conflicts can occur in a tank with only males of this species; they have also
observed interspecific sexual conflicts of Ginglymostoma cirratum
towards Carcharias taurus, which agrees with the findings of the present
study.
Gordon (1993) stated that clasper
flexion occurs in an elevated frequency when males start to perform
pre-copulatory behaviors and, based on the study of Myrberg & Gruber (1974)
with Sphyrna tiburo, stated the hypothesis that they might occur in
different times of the year as well.
Thus, this work presents not only the first evidence of clasper
movements in grey nurse sharks in a different context from mating, but also as
a new movement type.
In addition, our observations permit
us to hypothesize that the claspers’ displays are correlated with interspecific
sexual conflicts, especially as a response to aggressive behaviors performed by
the cohabitants, such as stalking, snapping and tailing. Probably, environmental stimuli present in
the tank initiate physiological responses such as clasper movements. Additional research on clasper movements,
however, is still needed. Studies in
different facilities and with bigger captive populations should be carried out
to permit a better understanding on this topic.
Another important question to
consider is if there are any steroid hormones influencing these behaviors,
since it is known that sexual conflicts are influenced by hormones, such as
testosterone and progesterone (Henningsen et al. 2008). Attention should be paid to the restricted
area in which such captive mature animals inhabit, which could affect the
hormones’ effects, since the most common tank has a semi-closed system (Mohan
& Aiken 2004) that may result in a concentration of these hormones.
Conclusions
Carcharias taurus can perform at least four clasper movements. Besides clasper flexion, splaying and
crossing, already described in the literature, this species can achieve a new mode,
clasper flaring. Although this behaviour
has already been seen in other species, this is the first report analyzing its
occurrence without a reproductive context.
The results suggest that clasper
movements are related to environmental stimuli, in a context different from
mating. We believe that the convergence
of reproductive behaviours of other species from the tank could have resulted
in a physiological response in the subject that affected its behaviour. This would explain not only why the clasper
movements are frequently seen in a captive context (since it normally has a
closed system), but also why the movements here described occurred together
with agonistic displays.
These results suggest the need to
explore water sample analyses to better understand the stimuli that are
released and that could affect the physiology behind behaviours and
interspecific relationships in natural and artificial environments. These analyses would also improve captive
husbandry techniques, since it could give us information on the need for
keeping species apart at certain periods to avoid interspecific conflicts in
confined environments.
References
Altmann, J. (1974). Observational study of behavior sampling methods. Behaviour
49: 227–267.
Barker, S.M., V.M.
Peddemors & J.E. Williamson (2011). A video and
photographic study of aggregation swimming and respiratory behavior changes in
the Grey Nurse Shark (Carcharias taurus) in response to the presence of
SCUBA divers. Marine and Freshwater Behaviour and Physiology 44: 75–92.
Carrier, J.C., H.L.
Pratt & L.K. Martin (1994). Group reproductive
behaviors in free-living Nurse Sharks, Ginglymostoma cirratum. Copeia
1994: 646–656.
Chapman, D.D., M.J.
Corcoran, G.M. Harvey, S. Malan & M.S. Shivji (2003). Mating behavior of southern Stingray, Dasyatis americana
(Dasyatidae). Environmental Biology of Fishes 68: 241–245.
Compagno, L.J.V.
(2001). Sharks of The World: An Annotated and
Illustrated Catalogue of Shark Species Known to Date: Bullhead, Mackerel and
Carpet Sharks (Heterodontiformes, Lamniformes and Orectolobiformes). FAO, Rome, 269pp.
Galván-Tirado, C., F. Galván-Magaña &
R.I. Ochoa-Báez (2014). Reproductive biology of the
Silky Shark Carcharhinus falciformis in the southern Mexican Pacific. Journal
of Marine Biological Association of the United Kingdom 95: 561–567.
Gilbert, P.W.
(1984). Biology and behavior of sharks. Endeavour
8: 179–187.
Gilbert, P.W. &
G.W. Heath (1972). The clasper-siphon sac mechanism in
Squalus acanthias and Mustelus canis. Comparative Biochemistry
and Physiology 42: 97–119.
Gordon, I. (1993). Pre-copulatory behavior of captive Sandtiger Sharks, Carcharias
taurus. Environmental Biology of Fishes 38: 159–164.
Hannon, G.E. &
A.C. Crook (2004). Observations on the behavior of
captive Sand Tiger Sharks, Carcharias taurus (Rafinesque, 1810), Pp.
51–61. In: Drum & Croaker. A Highly Irregular Journal for The Public
Aquarist. Special Edition No. 2. Elasmobranch Husbandry Symbosium, 217pp.
Henningsen, A.D.,
M.J. Smale, I. Gordon, R. Garner, R. Marin-Osorno & N. Kinnunen (2004). Captive breeding and sexual conflict in elasmobranchs. In: Smith,
M., D. Warmolts, D. Thoney & R. Hueter (eds.). The Elasmobranch
Husbandry Manual: Captive Care of Sharks, Rays and Their Relatives. Ohio
Biological Survey Inc., Ohio, 588pp.
Henningsen, A.D.,
F.L. Murru, L.E.L. Rasmussen, B.R. Whitaker & G.C. Violetta (2008). Serum levels of reproductive steroid hormones in captive Sand
Tiger Dharks, Carcharias taurus (Rafinesque), and comments on their
relation to sexual conflicts. Fish Physiology and Biochemistry 34:
437–446.
Klimley, A.P.
(1980). Observations of courtship and copulation in the
Nurse Shark, Ginglymostoma cirratum. Copeia 1980: 878–882.
Lehner, P.N.
(1996). Handbook of Ethological Methods. England: Cambridge University Press.
Luer, C.A. &
P.W. Gilbert (1985). Mating behavior, egg deposition,
incubation period, and hatching in the clearnose skate, Raja eglanteria.
Environmental Biology of Fishes 13: 161–171.
Martin, R.A.
(2007). A review of shark agonistic displays: comparison
of display features and implications for shark-human interactions. Marine
and Freshwater Behaviour and Physiology 40: 3–34.
McKibben, J.N.
& D.R. Nelson (1986). Patterns of movement and
grouping of Gray Reef Sharks, Carcharhinus amblyrhinchos, at Enewetak,
Marshall Islands. Bulletin of Marine Science 38: 89–110.
Mohan, P.J. &
A. Aiken (2004). Water quality and life support
systems for large elasmobranch exhibits. In: Smith, M., D. Warmolts, D. Thoney
& R. Hueter (eds.). The
Elasmobranch Husbandry Manual: Captive Care of Sharks, Rays and Their Relatives.
Ohio Biological Survey Inc., Ohio, 588pp.
Myrberg, A.A.,
& S.H. Gruber (1974). The behavior of the Bonnethead
Shark, Sphyrna tiburo. Copeia 1974: 358–374.
Powell, D.C., M.
Wisner & J. Rupp (2004). Design and
construction of exhibits for elasmobranchs. In: Smith, M., D. Warmolts, D.
Thoney & R. Hueter (eds.). The Elasmobranch Husbandry Manual: Captive
Care of Sharks, Rays and Their Relatives. Ohio Biological Survey Inc.,
Ohio, 588pp.
Pratt Jr, H.L.
& J.C. Carrier (2001). A review of elasmobranch
reproductive behavior with a case study on the Nurse Shark, Ginglymostoma
cirratum. Environmental Biology of Fishes 60: 157–188.
Ritter, E.K. &
L.V.J. Compagno (2013). Clasper flaring: maintenance behavior,
or a Normally Hidden Feature of Male Whitetip Reef Sharks, Triaenodon Obesus?
The Open Fish Science Journal 6: 10–12.
Tricas, T.C.
(1980). Courtship and mating-related behaviors in
Myliobatid rays. Copeia 1980: 553–556.
Uchida, S., M. Toda & Y. Kamei (1990). Reproduction of
elasmobranchs in captivity. In: Pratt Jr, H.L., S.H. Gruber & T. Taniuchi
(eds.). Elasmobranchs as Living Resources: Advances in The Biology, Ecology,
Systematics, and The Status of The Fisheries. NOAA Technical Report NMFS
90, Washington DC, 518pp.
Yano, K., F. Sato
& T. Takashi (1999). Observations of mating
behavior of the Manta Ray, Manta birostris, at the Ogasawara Islands,
Japan. Ichthyological Research 46: 289–296.