Journal of Threatened Taxa
| www.threatenedtaxa.org | 26 August 2018 | 10(9): 12173–12193
Diversity and status of avifauna in
man-made sacred ponds of Kurukshetra, India
Parmesh Kumar 1 & Archna Sharma 2
1,2 Department
of Zoology, University College, Kurukshetra
University, Thanesar, Kurukshetra,
Haryana 136119, India
1 parmeshkuk@rediffmail.com
(corresponding author), 2 sharma.archna6@gmail.com
doi: https://doi.org/10.11609/jott.3729.10.9.12173-12193 | ZooBank:
urn:lsid:zoobank.org:pub:AF8A19E1-DEFD-48B7-8221-7B7373C4F680
Editor: Hem S. Baral, Charles Sturt University, Albury-Wodonga, Australia. Date of publication: 26 August
2018 (online & print)
Manuscript details: Ms # 3729 | Received 12 August 2017 | Final received 07 August 2018
| Finally accepted 10 August 2018
Citation: Kumar, P. & A. Sharma (2018). Diversity and status of avifauna in
man-made sacred ponds of Kurukshetra, India. Journal of
Threatened Taxa 10(9): 12173–12193; https://doi.org/10.11609/jott.3729.10.9.12173-12193
Copyright: © Kumar & Sharma 2018. Creative
Commons Attribution 4.0 International License. JoTT
allows unrestricted use of this article in any medium, reproduction and
distribution by providing adequate credit to the authors and the source of
publication.
Funding: None.
Competing interests: The authors declare no competing interests.
Author Details: Dr. Parmesh Kumar is an
Associate Professor in Zoology, University College, Kurukshetra University Kurukshetra.
His field of research includes wildlife ecology and animal behaviour.
Ms. Archna
Sharma is a research scholar and pursuing her PhD from Department of
Zoology, Kurukshetra University Kurukshetra.
Author Contribution: PK and AS
conceived and designed the study. AS collected the field data and prepared rough
draft of the manuscript. PK guided the research, analyzed the data and wrote
the final draft of the manuscript.
Acknowledgements: We thank the Kurukshetra
University, Kurukshetra, for providing necessary
research facilities. We are grateful to B.L. Saini
for identification of flora and Omvir Singh for
preparing the map of the study area.
Abstract: Located in the Trans-Gangetic Plains of India,
Kurukshetra is dotted with a number of man-made,
perennial, sacred ponds of great historical and religious importance. These wetlands also serve as important
wintering and stopover sites for birds coming from the Palearctic region. Surveys were conducted from April 2014 to
March 2015 to record the diversity and status of avifauna in four sacred ponds of Kurukshetra. Point counts and direct observations were used
to record the bird species. A total of
126 bird species of 98 genera belonging to 45 families and 16 orders were
identified, of which 41 were winter migrants, six were summer migrants, and 79
were residents. Anatidae
(n=15) was the most common family, followed by Ardeidae
(n=8), and Motacillidae and Muscicapidae
(n=7 each). Based on the guilds, 37
species were carnivorous, 36 omnivorous, 29 insectivorous, six herbivorous, six
frugivorous, five granivorous,
four insectivorous/nectarivorous, and three piscivorous. Of the
species recorded, five species are classified as Near Threatened and one
species as Vulnerable in the IUCN Red List of Threatened Species; nine species
are listed in Appendix II of Convention on International Trade in Endangered
Species of Wild Fauna and Flora (CITES) and six species are included in
Schedule I of the Indian Wildlife (Protection) Act, 1972. We hope that this study will provide a
baseline for future research on monitoring the population and seasonal changes
in the bird assemblage of sacred ponds.
Keywords:
Avifauna, diversity, India, Kurukshetra, sacred
ponds, status.
INTRODUCTION
Wetlands are the most productive
biomes in the world (Kumar et al. 2005) and provide the transitional link
between aquatic and terrestrial habitats (Torell et
al. 2001; Zedler & Kercher
2005). They have specific ecological
characteristics, functions, and values, occupying about 6% of the earth’s
surface (Maltby & Turner 1983; Green 1996; Getzner 2002) and providing habitat to a wide array of
flora and fauna (Buckton 2007). Wetlands are, thus,
often considered as treasuries of biodiversity within a region or a landscape (Gopal & Sah 1993; Surana et al. 2007).
Birds constitute an important component of the biotic community of
wetland ecosystems as they occupy several trophic levels in the food web of
wetlands and form the terminal links in many aquatic food chains (Custer &
Osborn 1977). Because of their high
mobility, birds respond quickly to changes in their habitats (Morrison 1986);
they are, thus, valuable indicators of the ecological health, productivity,
trophic structure, human disturbance, and contamination of wetland ecosystems
(Custer & Osborn 1977; Subramanya 1996).
India, with its varied topography and climatic
regimes, supports diverse and unique wetland habitats that occupy an estimated
area of 15.26 million hectares (Panigrahy et al.
2012). Apart from natural wetlands, which support 20% of the known biodiversity
of India (Kumar et al. 2005), there are a large number of man-made wetlands
that also support rich flora and fauna. It is estimated that there are 5,55,557
small-sized wetlands (<2.25 ha) in the form of village tanks/ponds in India
(Panigrahy et al. 2012). These wetlands provide
suitable habitats and food resources for a wide variety of birds (Stewart 2007;
Ali et al. 2013). Of the 1,263 bird
species reported from India (Praveen et al. 2016), 310 species are known to be
dependent on wetlands (Kumar et al. 2005).
Wetlands in India, as elsewhere, however, are under tremendous
anthropogenic pressures including encroachment of wetland habitat,
unsustainable harvesting of resources, industrial pollution, poisoning,
agricultural runoff, eutrophication, siltation, and invasion of alien species
(Prasad et al. 2002). These impacts can lead to population declines and changes
in community structure of birds (Kler 2002; Verma et al. 2004; Reginald et al. 2007).
Biodiversity
inventories or checklists serve as repositories of baseline information on
species occurrences, biogeography, and their conservation status (Chandra &
Gajbe 2005).
They are essential tools for developing our knowledge and understanding
of biodiversity, and often the first step to evolve an appropriate long-term
conservation strategy for birds and their habitats (Kumar et al. 2005; Badola & Aitken 2010).
Located in the Trans-Gangetic Plains of India, the landscape of Kurukshetra is dotted with a number of perennial, man-made,
sacred wetlands of great historical and religious importance. A large number of pilgrims and
tourists visit these sacred tanks to take a holy dip and perform religious
ceremonies. These wetlands are also
potentially important for birds, not only because they provide foraging,
roosting, and breeding habitats for resident species, but also for their role
as stopover sites or wintering areas for several migrants of the Palearctic
region (Kumar et al. 2016). The avifauna
of these sacred wetlands, however, remains poorly known. Lack of adequate information on bird species
inhabiting wetlands greatly limits the development and establishment of
effective conservation strategies. The
present study was hence undertaken to make an inventory of bird species that
inhabit sacred ponds of Kurukshetra in the Trans-Gangetic Plains of India along with their conservation and
residential status.
MATERIALS AND METHODS
Study area
The present study was carried out in four religious ponds: namely,
Brahma Sarovar, Jyoti Sarovar, Baan Ganga, and Sannihit
Sarovar located in and around Kurukshetra
(29.866–30.200 0N & 76.416–77.066 0E), Haryana, in
the Trans-Gangetic Plains of India (Fig. 1, Table 1).
These ponds are surrounded by human habitations and
agricultural fields. The surrounding agriculture fields, with wheat and
paddy as main crops, provide extra foraging space and food for certain wetland
bird species. The study area,
experiencing sub-tropical climate, has three seasons: rainy (July–September),
cool-and-dry (October–February), and the hot-and-dry (March–June); temperature
ranges from 3–45 0C and annual rainfall averages to 582mm. The wetlands support many types of macrophytes that may be grouped into marginal, submerged,
floating, and emergent categories, of which Eichhornia
crassipes (a deadly invasive) is the dominant
free-floating, Hydrilla verticillata
the dominant submerged, and Cynodon dactylon the dominant marginal species in the wetlands.
Various tree species like Jamun Syzygium
cumini, Mango Mangifera
indica, Alstonia
sp., Acacia Acacia nilotica
& Acacia arabica, Neem
Azadirachta indica,
Jujube Zizyphus jujube, Wild Senna Cassia tora, Banyan
Tree Ficus benghalensis,
Bodhi Tree or Peepal Ficus
religiosa, and the Indian Rosewood Dalbergia sissoo at
the banks or in the surroundings of the ponds provide suitable roosting and
nesting sites for various bird species.
The ponds are also surrounded by Mesquite Prosopis
juliflora, a deadly invasive shrub, and the
non-native Eucalyptus sp.
Data collection
Bird surveys
were conducted at two-week intervals in all the ponds from April 2014 to March
2015, following the point count method (Bibby et al.
2000). Six to 10 vantage points, at
least 250m apart, were selected at the perimeter of each pond, and each point
location was surveyed 24 times during the entire study period. The observer waited for a few minutes after
arrival at each station before beginning to count. This allowed the birds to settle down following
the observer’s arrival and 10–20 minutes were spent at each point surveying the
birds. Birds were counted
directly, aided by 7x35 Nikon binoculars, during hours of peak activity
0600–1000 hr and 1600–1800 hr. In addition to these regular surveys,
opportunistic records were also collected during other time periods of the day
by walking at a slow pace along the bank of selected ponds and recording the
species observed. Field guides (Grimmett et al. 1999; Kazmierczak
& Perlo 2000) were used for field
identification. Taxonomy and
nomenclature follow Praveen et al. (2016).
For residential status, birds were categorised
as resident, winter visitor, and summer visitor on the basis of their presence
in the study area (Ali & Ripley 1987).
Feeding guilds were classified on the basis of direct observations and
available literature (Ali & Ripley 1987; Grimmett
et al. 1999). For national and global
conservation status of recorded avifauna, we followed IWPA (1972), CITES
(2012), and IUCN (2017). The relative
abundance (RA) of families was calculated using the following formula as per
Torre-Cuadros et al. (2007):
Number of species in a family
RA =
--------------------------------------------- x 100
Total number of species
Table 1. Summary of general characteristics of selected sacred ponds
|
|
Sacred
Pond |
Coordinates |
Elevation (m) |
General
Features |
|
1. |
Brahma Sarovar |
29.9500N
& 76.8160E |
245 |
Rich, healthy,
and robust perennial pond situated in the heart of Kurukshetra
city; one of largest man-made bathing tanks in Asia; divided into two sections,namely,
Eastern and Western Brahma Sarovar. The size of
Eastern and Western Brahma Sarovar is 548.64× 457.20×4.57
m and 457.20×457.20×4.57 m, respectively; edged with 6.09m wide platforms,
stairs, and a 12.19m wide parikrama. Bathing
platforms with protective railings have been constructed along the periphery
of the tank. Exclusive separate and covered bathing areas have been
constructed for use of women pilgrims. The water in the tank is replenished
using water from Bhakra irrigation canal. A large number of pilgrims and tourists take holy dips in the tank on auspicious days of the new moon and solar eclipse. To add scenic beauty, the sarovar is
decorated on the periphery with lush green lawns, floral beds, and huge trees with thick and dense canopy,
which serve as roosting and nesting sites for birds. |
|
2. |
Jyoti Sarovar |
29.9500N & 76.7660E |
253 |
Perennial,
a series of three closely located ponds at the outskirts of Jyotisar village; one
is used by the tourists and local people for holy dip. Size of Jyoti Sarovar is 393.7x196.8 x 3.7m. The second is used for
lotus cultivation, is mainly fed by direct precipitation and run-off from
surroundings, is recharged during summer through a feeder canal,and is
surrounded by large marshy swamp fed by local
village sewage; third is used for cattle drinking and bathing. Both the
second and third ponds are heavily infested with water hyacinth. |
|
3. |
Baan Ganga |
29.9330N & 76.8000E |
254 |
Perennial,
man-made, religious pond located at the outskirts of Dayalpur Village.
Size of of Baan Ganga is 258.20x127.6x 3.7m. It is
mainly fed by direct precipitation and run-off from surroundings, and is recharged during summer through field
channels. The tank is flanked by rural human habituations
and agricultural fields. In the vicinity of the sacred pond, there is a rural pond used for cattle
drinking and bathing, washing of vehicles, and other domestic purposes. |
|
4. |
Sannihit Sarovar |
29.9500N
& 76.8330E |
244 |
Perennial,
man-made sacred pond in the heart of Kurukshetra
city about 1km from Brahma Sarovar, 457.20x137.16 m
in size, surrounded by urban human habitation; used by pilgrims for bathing
and ‘pinddaan’. |
RESULTS AND DISCUSSION
A total of 126 species
of birds belonging to 98 genera, distributed among 45 families and 16 orders were
recorded from four sacred ponds of Kurukshetra during
the study period (Table 2, Images 1–102).
Of these, 62 species were wetland-associated and the rest were
terrestrial. Of all species recorded, 31
(24.60%) were observed from all the four sacred wetlands, and 95 (75.39%) were
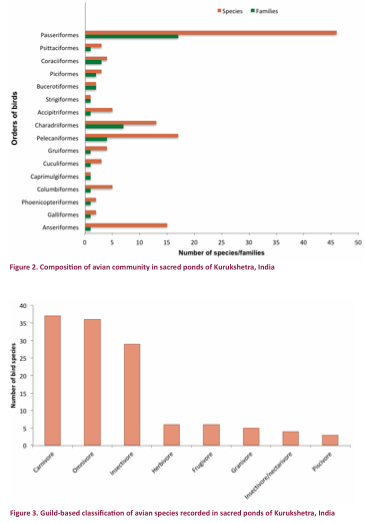
recorded from some specific wetlands alone (Table 2). Passeriformes had the highest diversity with
46 species and 17 families (Fig. 2). The
proportion of species richness of birds by family varied from 0.79–11.90%. Anatidae, the
richest family represented by 15 species, accounted for 11.90% of the total
bird species in the study area (Table 3).
Apodidae, Burhinidae,
Rostratulidae, Strigidae, Bucerotidae, Upupidae, Picidae, Meropidae, Coraciidae, Campephagidae, Dicruridae, Nectariniidae, Ploceidae, Passeridae, Pycnonotidae, Zosteropidae, and Timaliidae were poorly represented families with a single
species in each. Similarly, Gupta & Kumar(2009) recorded 110 bird species belonging to 41
families and 14 orders from different habitats of Kurukshetra. For comparison, Alfred et al. (2001) reported
216 wetland bird species from various wetland habitats in the much more
expansive Sub-Himalayan Terai and Indo-Gangetic Plains of northern India. Ducks and geese (Anatidae)
are the most copious and remarkable winter migrants to the Indian-subcontinent,
and constitute about 85% of the migrant bird populations (Alfred et al.
2001). These results are in confirmation
with findings of earlier workers who have reported Anatidae
to be the most dominant family among bird communities in different wetland
habitats of Haryana in northern India (Kumar & Gupta 2009, 2013; Tak et al. 2010; Kumar et al. 2016).
Of the 126 species identified, 41 were winter migrants, six were
summer migrants, and 79 were residents.
The occurrence of a considerable number of winter migratory species can
be attributed partly to the study area being on the Central Asian Flyway and
serving as a wintering and stopover site for migratory birds that breed in the
Palearctic region (Kumar et al. 2016).
These migratory birds form a major component of the aquatic bird
populations in various wetland habitats of northern India (Alfred et al. 2001; Manral et al. 2013; Kumar et al. 2016). We observed that the majority of the winter migrants
stayed in the sacred wetlands from November to February. The summer visitors, including Cotton Teal Nettapus coromandelianus,
Lesser Whistling Duck Dendrocygna javanica, Comb Duck Sarkidiornis
melanotos, Pied Cuckoo Clamator
jacobinus, Pheasant-tailed Jacana Hydrophasianus chirurgus,
and Greater Painted-snipe Rostratula benghalensis were spotted during summer season
(April–August) in the study area. Little
Cormorant Microcarbo niger and Black-winged Stilt Himantopus
himantopus, being common resident species, were
recorded in and around the wetlands throughout the year, but their populations
augmented due to the influx of migrant birds during the winter season.
Wetland characteristics like size, water depth, quality
of water, trophic structure, and presence of suitable roosting and nursery
sites influence the abundance and diversity of birds (Wiens
1989; Mukherjee et al. 2002; Ma et al. 2010).
During the study period, species richness was recorded to be the highest
at Jyoti Sarovar (n=107),
followed by Brahma Sarovar (n=88), Baan Ganga (n=53),
and Sannihit Sarovar
(n=34). Generally, habitats with a
complex architecture generate greater resources for birds, allowing the
persistence of a greater number of species and guilds than in less complex
habitats (MacArthur & MacArthur 1961; Tews et al.
2004; Codesido et al. 2013). In the present study, Jyoti
Sarovar wetland, along with the adjacent rural pond,
marshy area, and irrigated crop fields, provided a mosaic of habitats leading
to multiple and variety of alternative food sources for the birds, and thus
registered highest species richness (Aynalem & Bekele 2008). Brahma
Sarovar and Sannihit Sarovar, being located in urban areas of the Kurukshetra City, are more exposed to local people and
tourists. As a result, bird activities
like feeding, nesting, hiding, and breeding are affected at these sites.
The quality and quantity
of food available is the major factor that determines the spatio-temporal distribution and relative abundance of
birds in a given habitat (Wiens 1989; Ma et al. 2010;
Jha 2013). The
different species of birds occupying a particular feeding guild and space have
evolved specialized foraging strategies to explore and obtain food resources
efficiently and thereby to reduce competition (Nudds
& Bowlby 1984).
As far as foraging habits of the bird community in the study area are
concerned, eight major feeding guilds were identified (Fig. 3). This representation of major trophic guilds
in the area indicated that the area holds a wide spectrum of food resources for
birds. The carnivore guild was the most
abundant one with 37 species followed by omnivore (36), insectivore (29),
herbivore (six), frugivore (six), granivore
(five), insectivore/nectarivore (four), and piscivore (three).
Due to their specialized diet and low availability of preferable food
resources, the nectarivores and piscivores
are generally less represented (Wiens 1989). The diversity of avifauna in the study area
may be due to the presence of a wide spectrum of food niches, which reduced
food competition among different species (Jose & Zacharias 2003). About half of the recorded bird species were
those associated with wetland habitats, such as ducks, herons, egrets,
cormorants, jacanas, grebes, kingfishers, and storks, which were observed to
feed on aquatic organisms (worms, insects, snails, fish, and amphibians) at
various water depths available in the wetlands and adjoining paddy fields and
marshy area.
Bronze-winged Jacana Metopidius
indicus and Pheasant-tailed Jacana Hydrophasianus chirurgus
were spotted at Jyoti Sarovar
alone, the only pond with lotuses. The
vegetation cover of lotuses provides suitable feeding, nesting, and breeding
habitat for herons, moorhens, and jacanas.
Purple Swamphen Porphyrio
porphyrio, a common resident species, was
observed only in weedy marsh areas flanking the sacred pond of Jyoti Sarovar, where there were
frequent human activities; this bird species may be a bio-indicator of enhanced
weed infestation and increased vegetation cover in the wetlands of Haryana
(Kumar et al. 2016). Waders, shorebirds,
Purple Moorhen, and wagtails were also observed foraging in the irrigated wheat
and paddy fields flanking the sacred ponds in rural habitats (Jyoti Sarovar and Baan
Ganga). This observation is consistent
with earlier reports, where foraging by aquatic birds outside the wetlands in
surrounding agriculture fields has been recorded (Lane & Fujioka 1998;
Mukherjee et al. 2002; Urfi 2003; Jha
2013; Kumar et al. 2016).
Among the recorded
avifauna, five species, namely, Painted Stork Mycteria
leucocephala,
Black-necked Stork Ephippiorhynchus asiaticus, Black-headed Ibis Threskiornis
melanocephalus, River Tern Sterna aurantia, and Alexandrine Parakeet Psittacula
eupatria are listed as Near Threatened, and one
species, Common Pochard Aythya
ferina, as Vulnerable in the IUCN Red List (IUCN
2017). All the remaining species (n=120)
are placed in the Least Concern category in the Red List of IUCN (2017). Additionally, nine species are included in
Appendix-II of CITES (2012). Six
species, including five species of Accipitridae and
one of Phasianidae, are considered nationally
threatened as these are listed under Schedule-I of the Indian Wildlife
(Protection) Act, 1972.
Significant records
Painted Stork - Near
Threatened: A winter migrant in the study area was recorded in a small
flock (4–8 individuals) only at Jyoti Sarovar during winter months (December–January). The birds were often recorded roosting on
large trees at the bank of the wetland.
Black-necked Stork - Near Threatened: A lone male individual was
recorded foraging in the mud-flat adjacent to Jyoti Sarovar on 25January
2015. This species is very widely but
thinly distributed in India, with the northern and northwestern regions forming
its main strongholds (Rahmani 1989).
Black-headed Ibis - Near Threatened: A resident wader species
(Kumar et al. 2016) was recorded in small loose groups (1–4 individuals) only
at Jyoti Sarovar throughout
the study period. It was often observed
foraging with other waders at the margins of the pond, and mudflats and paddy
fields adjoining the sacred wetland.
River Tern - Near Threatened: A common resident species in the
study area (Kumar et al. 2016) was recorded as 1–7 scattered individuals at all
the four sacred ponds throughout the study period.
Alexandrine Parakeet - Near Threatened: A resident species in the
study area was recorded in small groups of 5–10 individuals. The birds were frequently observed roosting
on trees at banks of all the ponds.
Common Pochard - Vulnerable: This is a
common winter visitor in India (Grimmett et al.
1999). The species was recorded in flocks of 6–50 individuals during winter
months (November–March) in Brahma Sarovar only.
Comb Duck - Appendix II of CITES: A resident species in the Indian
subcontinent with local movements (Grimmett et al.
1999) was recorded only at Jyoti Sarovar
in a pair during summer (May 2014).
In addition to the cultural and religious legacy of the region,
the presence of significant numbers of migratory species as well as those with
conservation priorities underlines the importance of these sacred wetlands as
important bird habitats in Haryana. It
is evident from the present study that if some attention is provided to these
sacred wetlands, these could be developed as a good site for harbouring avifauna and as a haven for bird-watchers. Our efforts contributed towards filling
biological information gaps in the region; continuing studies will allow
monitoring of the population and seasonal changes in the bird assemblage.
Table 2. Checklist and status of avifauna recorded in sacred ponds of Kurukshetra in the Trans-Gangetic
Plains, India
Order/family/common name Scientific name Residential Feeding Conservation
status Sacred pond Image
atatus
guild IUCN CITES
IWPA
(2017)
(2012) (1972) BS
JS BG SS
ANSERIFORMES
Anatidae (15)
1 Lesser Whistling Duck Dendrocygna javanica (Horsfield, 1821) SM O LC - IV û ü û û 1
2 Common Shelduck Tadorna tadorna (Linnaeus, 1758) WM O LC - IV ü û û û
3 Ruddy Shelduck Tadorna ferruginea (Pallas, 1764) WM O LC - IV ü û û û 2
4 Red Crested Pochard Netta rufina (Pallas, 1773) WM H LC - IV ü û û û 3
5 Common Pochard Aythya ferina (Linnaeus, 1758) WM O VU - IV ü û û û 4
6 Tufted Duck Aythya fuligula (Linnaeus, 1758) WM H LC - IV ü û û û 5
7 Northern Shoveler Spatula clypeata (Linnaeus, 1758) WM O LC - IV ü ü û û 6
8 Gadwall Mareca strepera (Linnaeus, 1758) WM H LC - IV ü ü û û 7
9 Eurasian Wigeon Mareca penelope (Linnaeus,
1758) WM H LC - IV ü û û û
10 Indian Spot-billed Duck Anas poecilorhyncha Forster, 1781 WM H LC - IV ü ü û û 8
11 Mallard Anas platyrhynchos Linnaeus, 1758 WM H LC - IV ü ü û û 9
12 Northern Pintail Anas acuta Linnaeus, 1758 WM O LC - IV ü û û û 10
13 Common Teal Anas crecca Linnaeus, 1758 WM O LC - IV ü ü û û 11
14 Comb Duck Sarkidiornis melanotos (Pennant, 1769) SM O LC II IV û ü û û 12
15 Cotton Teal Nettapus coromandelianus (Gmelin, 1789) SM O LC - IV û ü û û 13
GALLIFORMES
Phasianidae (2)
16 Indian Peafowl Pavo cristatus Linnaeus, 1758 R O LC - I ü û û û 14
17 Grey Francolin Francolinus pondicerianus (Gmelin, 1789) R O LC - IV ü ü û û
PHOENICOPTERIFORMES
Podicipedidae (2)
18 Little Grebe Tachybaptus ruficollis (Pallas, 1764) R C LC - IV ü ü ü û 15
19 Great Crested Grebe Podiceps cristatus (Linnaeus, 1758) WM C LC - IV ü û û û 16
COLUMBIFORMES
Columbidae (5)
20 Rock Pigeon Columba livia Gmelin, 1789 R G LC - IV ü ü ü ü 17
21 Spotted Dove Spilopelia chinensis (Scopoli, 1786) R G LC - IV ü ü ü ü 18
22 Eurasian Collared Dove Streptopelia decaocto Frivaldszky, 1838 R G LC - IV ü ü ü ü 19
23 Laughing Dove Spilopelia senegalensis (Linnaeus, 1766) R G LC - IV ü ü ü û 20
24 Yellow-legged Green Pigeon Treron phoenicopterus (Latham, 1790) R F LC - IV ü ü ü ü 21
CAPRIMULGIFORMES
Apodidae (1)
25 Indian House Swift Apus affinis (Gray, 1830) R In LC - IV û ü ü û
CUCULIFORMES
Cuculidae (3)
26 Pied Cuckoo Clamator jacobinus (Boddaert, 1783) SM In LC - IV ü û ü û
27 Asian Koel Eudynamys scolopaceus (Linnaeus, 1758) R O LC - IV ü ü ü ü 22
28 Greater Coucal Centropus sinensis (Stephens, 1815) R C LC - IV ü ü ü ü 23
GRUIFORMES
Rallidae (4)
29 White-breasted Waterhen Amaurornis phoenicurus (Pennant, 1769) R O LC - IV ü ü ü ü 24
30 Purple Swamphen Porphyrio porphyrio (Linnaeus, 1758) R O LC - IV û ü û û 25
31 Common Moorhen Gallinula chloropus (Linnaeus, 1758) WM O LC - IV ü ü ü ü 26
32 Common Coot Fulica atra Linnaeus, 1758 WM O LC - IV ü ü ü ü 27
PELECANIFORMES
Ciconiidae (3)
33 Painted Stork Mycteria leucocephala (Pennant, 1769) WM C NT - IV û ü û û 28
34 Asian Openbill Anastomus oscitans (Boddaert, 1783) WM C LC - IV û ü û û 29
35 Black-necked Stork Ephippiorhynchus asiaticus (Latham, 1790) WM C NT - IV û ü û û
Ardeidae (8)
36 Black-crowned Night-Heron Nycticorax nycticorax (Linnaeus, 1758) R C LC - IV ü ü ü û 30
37 Indian Pond Heron Ardeola grayii (Sykes, 1832) R C LC - IV ü ü ü ü 31
38 Cattle Egret Bubulcus ibis (Linnaeus,
1758) R C LC - IV ü ü ü ü 32
39 Grey Heron Ardea cinerea Linnaeus, 1758 R C LC - IV û ü û û 33
40 Purple heron Ardea purpurea Linnaeus, 1766 R C LC - IV ü ü û û 34
41 Great Egret Ardea alba Linnaeus, 1758 WM C LC - IV ü ü ü û 35
42 Intermediate Egret Ardea intermedia Wagler, 1829 WM C LC - IV ü ü ü û 36
43 Little Egret Egretta garzetta (Linnaeus, 1766) R C LC - IV ü ü ü ü 37
Threskiornithidae (3)
44 Black-headed Ibis Threskiornis melanocephalus (Latham, 1790) R C NT - IV û ü û û 38
45 Indian Black Ibis Pseudibis papillosa (Temminck, 1824) R C LC - IV û ü û û 39
46 Glossy Ibis Plegadis falcinellus (Linnaeus, 1766) R C LC - IV û ü û û 40
Phalacrocoracidae (3)
47 Little Cormorant Microcarbo niger
(Vieillot, 1817) R C LC - IV ü ü ü ü 41
48 Great Cormorant Phalacrocorax carbo (Linnaeus, 1758) WM C LC - IV ü ü û ü 42
49 Indian Cormorant Phalacrocorax fuscicollis Stephens, 1826 WM P LC - IV ü ü û ü 43
CHARADRIIFORMES
Burhinidae (1)
50 Eurasian Thick-knee Burhinus oedicnemus (Linnaeus, 1758) R O LC - IV û ü û û 44
Recurvirostridae (2)
51 Pied Avocet Recurvirostra avosetta Linnaeus, 1758 WM C LC - IV û ü û û 45
52 Black-winged stilt Himantopus himantopus (Linnaeus, 1758) R C LC - IV ü ü ü û 46
Charadriidae (2)
53 Red-wattled Lapwing Vanellus indicus (Boddaert, 1783) R C LC - IV ü ü ü ü 47
54 White-tailed Lapwing Vanellus leucurus (Lichtenstein,
1823) WM C LC - IV û ü û û 48
Rostratulidae (1)
55 Greater Painted-Snipe Rostratula benghalensis (Linnaeus, 1758) SM O LC - IV û ü û û
Jacanidae (2)
56 Pheasant-tailed Jacana Hydrophasianus chirurgus (Scopoli, 1786) SM O LC - IV û ü û û 49
57 Bronze-winged Jacana Metopidius indicus (Latham,
1790) R O LC - IV û ü û û 50
Scolopacidae (3)
58 Common Sandpiper Actitis hypoleucos Linnaeus, 1758 WM C LC - IV ü ü û û 51
59 Common Greenshank Tringa nebularia (Gunnerus, 1767) WM C LC - IV û ü û û 52
60 Common Redshank Tringa totanus (Linnaeus, 1758) WM C LC - IV û ü û û 53
Laridae (2)
61 Pallas’s Gull Larus ichthyaetus Pallas,
1773 WM C LC - IV ü û û û
62 River Tern Sterna aurantia Gray, 1831 R P NT - IV ü ü û û 54
ACCIPITRIFORMES
Accipitridae (5)
63 Black-winged Kite Elanus caeruleus (Desfontaines, 1789) R C LC II I û ü û ü 55
64 Oriental Honey Buzzard Pernis ptilorhynchus (Temminck, 1821) R C LC II I ü ü û û
65 Shikra Accipiter badius (Gmelin, 1788) R C LC II I ü ü ü û 56
66 Brahminy Kite Haliastur Indus (Boddaert, 1783) R C LC II I ü ü û û 57
67 Black Kite Milvus migrans (Boddaert, 1783) R C LC II I ü ü ü ü 58
STRIGIFORMES
Strigidae (1)
68 Spotted Owlet Athene brama (Temminck, 1821) R C LC II IV ü ü ü û 59
BUCEROTIFORMES
Bcerotidae (1)
69 Indian Grey Hornbill Ocyceros birostris (Scopoli, 1786) R O LC - IV ü ü ü ü 60
Upupidae (1)
70 Common Hoopoe Upupa epops Linnaeus, 1758 R In LC - IV û ü ü û 61
PICIFORMES
Picidae (1)
71 Lesser Golden-backed Woodpecker Dinopium benghalense (Linnaeus,
1758) R In LC - IV ü ü û û 62
Ramphastidae (2)
72 Brown-headed Barbet Psilopogon zeylanicus (Gmelin, 1788) R F LC - IV ü ü ü û 63
73 Coppersmith Barbet Psilopogon haemacephalus (Muller, 1776) R F LC - IV ü ü û û 64
CORACIIFORMES Meropidae (1)
74 Green Bee-eater Merops orientalis Latham, 1802 R In LC - IV ü ü ü ü 65
Coraciidae (1)
75 Indian Roller Coracias benghalensis (Linnaeus, 1758) R C LC - IV û ü ü û 66
Alcedinidae (2)
76 Pied Kingfisher Ceryle rudis (Linnaeus, 1758) R P LC - IV ü û û û
77 White-throated Kingfisher Halcyon smyrnensis (Linnaeus, 1758) R C LC - IV ü ü ü ü 67
PSITTACIFORMES
Psittaculidae (3)
78 Slaty-headed Parakeet Psittacula himalayana (Lesson, 1832) WM F LC II IV ü û û û
79 Alexandrine Parakeet Psittacula eupatria (Linnaeus,
1766) R F NT II IV ü ü ü ü 68
80 Rose-ringed Parakeet Psittacula krameri (Scopoli, 1769) R F LC - IV ü ü ü ü 69
PASSERIFORMES Campephagidae
(1)
81 Scarlet Minivet Pericrocotus flammeus (Forster, 1781) WM In LC - IV ü û û û
Dicruridae (1)
82 Black Drongo Dicrurus macrocercus Vieillot, 1817 R In LC - IV ü ü ü ü 70
Laniidae (2)
83 Bay-backed Shrike Lanius vittatus Valenciennes, 1826 R C LC - IV û ü û û
84 Long-tailed Shrike Lanius schach Linnaeus, 1758 R C LC - IV û ü û û 71
Corvidae (3)
85 Rufous Treepie Dendrocitta vagabunda (Latham, 1790) R O LC - IV ü ü ü ü 72
86 House Crow Corvus splendens Vieillot, 1817 R O LC - V ü ü ü ü 73
87 Large-billed Crow Corvus macrorhynchos Wagler, 1827 WM O LC - IV ü ü ü ü 74
Nectariniidae (1)
88 Purple Sunbird Cinnyris asiaticus (Latham, 1790) R In/N LC - IV ü ü ü ü 75
Ploceidae (1)
89 Baya Weaver Ploceus philippinus (Linnaeus, 1766) R O LC - IV ü ü ü û 76
Estrildidae (2)
90 Indian Silverbill Euodice malabarica (Linnaeus, 1758) R G LC III IV û ü û û 77
91 Scaly-breasted Munia Lonchura punctulata (Linnaeus, 1758) R O LC - IV ü ü û û 78
Passeridae (1)
92 House Sparrow Passer domesticus (Linnaeus, 1758) R O LC - IV û ü ü û 79
Motacillidae (7)
93 Tree Pipit Anthus trivialis (Linnaeus, 1758) WM In LC - IV û ü û û
94 Paddyfield Pipit Anthus rufulus Vieillot, 1818 R In LC - IV û ü û û 80
95 Western Yellow Wagtail Motacilla flava Linnaeus,
1758 WM In LC - IV û ü û û 81
96 Grey Wagtail Motacilla cinerea Tunstall, 1771 WM In LC - IV û ü û û 82
97 Citrine Wagtail Motacilla citreola Pallas, 1776 WM In LC - IV û ü û û 83
98 White-browed Wagtail Motacilla maderaspatensis Gmelin, 1789 R In LC - IV ü ü ü ü
99 White Wagtail Motacilla alba Linnaeus, 1758 WM In LC - IV ü ü ü ü 84
Cisticolidae (4)
100 Zitting Cisticola Cisticola juncidis (Rafinesque, 1810) R In LC - IV ü ü ü û
101 Ashy Prinia Prinia socialis Sykes, 1832 R In/N LC - IV ü ü ü û 85
102 Plain Prinia Prinia inornata Sykes, 1832 R In LC - IV û ü û û 86
103 Common Tailorbird Orthotomus sutorius (Pennant, 1769) R In/N LC - IV ü ü ü û 87
Hirundinidae (6)
104 Northern House Martin Delichon urbicum (Linnaeus, 1758) R In LC - IV û ü û û
105 Wire-tailed Swallow Hirundo smithii Leach, 1818 R In LC - IV ü ü û û 88
106 Barn Swallow Hirundo rustica Linnaeus, 1758 R In LC - IV ü û û û
107 Eurasian Crag-Martin Ptyonoprogne rupestris (Scopoli, 1769) R In LC - IV ü û û û
108 Plain Martin Riparia paludicola (Vieillot, 1817) R In LC - IV ü û û û
109 Sand Martin Riparia riparia (Linnaeus, 1758) R In LC - IV ü û û û
Pycnonotidae (1)
110 Red vented Bulbul Pycnonotus cafer (Linnaeus, 1766) R O LC - IV ü ü ü ü 89
Zosteropidae (1)
111 Oriental White-eye Zosterops palpebrosus (Temminck, 1824) R In/N LC - IV ü ü û û 90
Timaliidae (1)
112 White-browed Scimitar Babbler Pomatorhinus schisticeps Hodgson, 1836 WM O LC - IV û ü û û
Leiothrichidae (3)
113 Large Grey Babbler Argya malcolmi (Sykes, 1832) R O LC - IV ü ü û û 91
114 Common Babbler Argya caudata (Dumont, 1823) R O LC - IV ü ü ü ü
115 Jungle Babbler Turdoides striata (Dumont, 1823) R O LC - IV ü ü û û 92
Sturnidae (4)
116 Asian Pied Starling Gracupica contra (Linnaeus,
1758) R O LC - IV ü ü ü û 93
117 Brahminy Starling Sturnia pagodarum ( Gmelin, 1789) R O LC - IV ü ü ü û 94
118 Common Myna Acridotheres tristis (Linnaeus, 1766) R O LC - IV ü ü ü ü 95
119 Bank Myna Acridotheres ginginianus (Latham, 1790) R O LC - IV ü ü ü û 96
Muscicapidae (7)
120 Indian Robin Saxicoloides fulicatus (Linnaeus, 1766) R In LC - IV ü ü ü û 97
121 Oriental Magpie Robin Copsychus saularis (Linnaeus,
1758) R In LC - IV ü ü ü ü 98
122 Verditer Flycatcher Eumyias thalassinus Swainson, 1838 WM In LC - IV ü û û û
123 Bluethroat Cyanecula svecica (Linnaeus, 1758) WM In LC - IV û ü û û 99
124 Red-breasted Flycatcher Ficedula parva (Bechstein, 1792) WM In LC - IV û ü û û 100
125 Common Stonechat Saxicola torquatus (Linnaeus, 1766) WM In LC - IV û ü û û 101
126 Brown Rock Chat Oenanthe fusca (Blyth, 1851) R In LC - IV ü ü û û 102
IUCN: International
Union for Conservation of Nature and Natural Resources; CITES: Convention on
International Trade in Endangered Species of Wild Fauna and Flora; IPWA: Indian
Wildlife Protection Act; R: Resident; SM: Summer migrant; WM: Winter migrant; LC:
Least concern species; NT: Near threatened species; VU: Vulnerable species; I:
Schedule- I species of IWPA (high priority species); IV: Schedule - IV species
of IWPA (relatively low priority species); BS - Brahma Sarovar;
JS - Jyoti Sarovar; BG -
Baan Ganga, Dayalpur; SS - Sannihit
Sarovar; C-Carnivore; H-Herbivore; In - Insectivore;
O - Omnivore; N - Nectarivore; F - Fruigivore; G - Grainivore; P - Piscivore; ü - Species recorded in the habitat; û - Species not recorded in the habitat.
Table 3. Relative diversity of various avian families in sacred ponds of
Kurukshetra, India
|
Order |
Family |
No. of species recorded |
Relative
abundance (%) |
|
Anseriformes |
Anatidae |
15 |
11.90 |
|
Galliformes |
Phasianidae |
2 |
1.59 |
|
Phoenicopteriformes |
Podicipedidae |
2 |
1.8 |
|
Columbiformes |
Columbidae |
5 |
3.96 |
|
Caprimulgiformes |
Apodidae |
1 |
0.79 |
|
Cuculiformes |
Cuculidae |
3 |
2.38 |
|
Gruiformes |
Rallidae |
4 |
3.17 |
|
Pelecaniformes |
Ciconiidae |
3 |
2.38 |
|
|
Ardeidae |
8 |
6.34 |
|
|
Threskiornithidae |
3 |
2.38 |
|
|
Phalacrocoracidae |
3 |
2.38 |
|
Charadriiformes |
Burhinidae |
1 |
0.79 |
|
|
Recurvirostridae |
2 |
1.59 |
|
|
Charadriidae |
2 |
1.59 |
|
|
Rostratulidae |
1 |
0.79 |
|
|
Jacanidae |
2 |
1.59 |
|
|
Scolopacidae |
3 |
2.38 |
|
|
Laridae |
2 |
1.59 |
|
Accipitriformes |
Accipitridae |
5 |
3.96 |
|
Strigiformes |
Strigidae |
1 |
0.79 |
|
Bucerotiformes |
Bucerotidae |
1 |
0.79 |
|
|
Upupidae |
1 |
0.79 |
|
Piciformes |
Picidae |
1 |
0.79 |
|
|
Ramphastidae |
2 |
1.59 |
|
Coraciiformes |
Meropidae |
1 |
0.79 |
|
|
Coraciidae |
1 |
0.79 |
|
|
Alcedinidae |
2 |
1.59 |
|
Psittaciformes |
Psittaculidae |
3 |
2.38 |
|
Passeriformes |
Campephagidae |
1 |
0.79 |
|
|
Dicruridae |
1 |
0.79 |
|
|
Laniidae |
2 |
1.59 |
|
|
Corvidae |
3 |
2.38 |
|
|
Nectariniidae |
1 |
0.79 |
|
|
Ploceidae |
1 |
0.79 |
|
|
Estrildidae |
2 |
1.59 |
|
|
Passeridae |
1 |
0.79 |
|
|
Motacillidae |
7 |
5.55 |
|
|
Cisticolidae |
4 |
3.17 |
|
|
Hirundinidae |
6 |
4.76 |
|
|
Pycnonotidae |
1 |
0.79 |
|
|
Zosteropidae |
1 |
0.79 |
|
|
Timaliidae |
1 |
0.79 |
|
|
Leiothrichidae |
3 |
2.38 |
|
|
Sturnidae |
4 |
3.17 |
|
|
Muscicapidae |
7 |
5.55 |
REFERENCES
Alfred,
J.R.B., A. Kumar, P.C. Tak & J.P. Sati (2001). Waterbirds of Northern India. Zoological
Survey of India, Kolkata, xxvi+468pp.
Ali, S. & S.D. Ripley (1987). Compact Handbook of the Birds of India and Pakistan together
with those of Bangladesh, Nepal, Bhutan and Sri Lanka. Oxford University Press, Delhi, 737pp.
Ali, A.M.S., S.R. Kumar & P.R. Arun
(2013). Waterbird assemblage in rural ponds of Samakhiali
region, Kutch District, Gujarat, India. Bird
Populations 12: 12–18.
Aynalem, S. & A. Bekele
(2008). Species composition, relative abundance and
distribution of bird fauna of riverine and wetland habitats of Infranz and Yiganda at southern tip of Lake Tana, Ethiopia. Tropical
Ecology 49(2): 199–209.
Badola, H.K. &
S. Aitken(2010). Biological
resources and poverty alleviation in the Indian Himalayas. Biodiversity
11(3&4): 8–18; http://doi.org/10.1080/14888386.2010.9712659
Bibby, C.J., N.D.
Burgess, D.A. Hill & S.H. Mustoe (2000). Bird Cen-sus Techniques. Academic Press, London, 302pp.
Buckton, S. (2007). Managing wetlands for sustainable
livelihoods at Koshi Tappu.
Danphe 16(1): 12–13.
Chandra, K. & P.U. Gajbe (2005). An inventory of herpetofauna of Madhya Pradesh and Chhattisgarh. Zoos’
Print Journal 20(3): 1812–1819; http://doi.org/10.11609/JoTT.ZPJ.1087.1812-9
CITES (2012). Checklist of Convention on International Trade
in Endangered Species of Wild Fauna and Flora. CITES, Geneva, Switzerland.
Downloaded on 22 December 2016; http://www.cites.org
Codesido,M., C.M.
González-Fischer & D.N. Bilenca (2013). Landbird assemblages in different agricultural
landscapes: a case study in the Pampas of Central Argentina. The Condor 115(1):
8–16; http://doi.org/10.1525/cond.2012.120011
Custer, T.W. & R.G. Osborn
(1977). Wading birds as biological indicators: 1975 colony survey. United States fish and wildlife Services, Special Scientific
Report-Wildlife. No. 206.
Getzner, M. (2002).
Investigating public decisions about protecting wetlands. Journal of
Environmental Management 64(3): 237–246; http://doi.org/10.1006/jema.2001.0471
Gopal, B. & M. Sah (1993).
Conservation and management of rivers in India: case study of the River Yamuna.
Environmental Conservation 20(3):243–254; http://doi.org/10.1017/S0376892900023031
Green, A.J. (1996). Analysis of
globally threatened Anatidae in relation to threats,
distribution, migration patterns, and habitat use. Conservation Biology 10(5): 1435–1445; http://doi.org/10.1046/j.1523-1739.1996.10051435.x
Grimmett, R., C. Inskipp & T. Inskipp (1999). Pocket Guide to the Birds of the Indian Subcontinent. Oxford University Press,
New Delhi, India, 384pp.
Gupta, S.K. & P.
Kumar (2009). Survey of avian fauna in and around Kurukshetra, Haryana,
India. The Indian Forester 135(10): 1367–1376.
IUCN (2017). The IUCN Red List of Threatened Species, Version 2017-1. Downloaded
on 20 August 2017; www.iucnredlist.org
IWPA (1972). The Indian Wildlife (Protection)
Act, 1972 (as amended upto 1993).Ministry of Environment, Forest and Climate Change,
Government of India, Delhi. Downloaded on 04 April 2017; http://www.envfor.nic.in/legis/wildlife/wildlife1
Jha,
K.K. (2013). Aquatic food plants and their consumer
birds at Sandi Bird Sanctuary, Hardoi, Northern
India. Asian Journal of Conservation Biology
2(1): 30–43.
Jose, B. & V.J. Zacharias
(2003). Distribution of birds in relation to vegetation
in the Calicut University Campus, Kerala. Zoos’ Print Journal
18(9): 1187–1192; http://doi.org/10.11609/JoTT.ZPJ.18.9.1187-92
Kazmierczak, K. & B.V. Perlo (2000). A Field Guide to the Birds of India, Sri Lanka, Pakistan, Nepal,
Bhutan, Bangladesh and the Maldives. Om Book Service, New Delhi, 352pp.
Kler, T.K. (2002). Bird
species in Kanjali wetland.
Tiger Paper 39(1): 29–32.
Kumar, P. & S.K.
Gupta (2009). Diversity and abundance of wetland birds
around Kurukshetra, India. Our
Nature 7(1): 212–217; http://doi.org/10.3126/on.v7i1.2574
Kumar, P. & S.K.
Gupta (2013). Status of wetland birds
of Chhilchhila Wildlife Sanctuary, Haryana, India. Journal of
Threatened Taxa 5(5):
3469–3976; http://doi.org/10.11609/JoTT.o3158.3969-76
Kumar, A., J.P. Sati, P.C. Tak & J.R.B. Alfred (2005). Handbook on Indian Wetland Birds
and their Conservation. Zoological Survey of
India, Kolkata,India, xxvi+468pp.
Kumar, P., D. Rai & S.K. Gupta (2016). Wetland bird assemblage
in rural ponds of Kurukshetra,
India. Waterbirds 39(1): 86–98; http://doi.org/10.1675/063.039.0111
Lane, S.J. & M. Fujioka (1998). The impact of changes in irrigation practices on the
distribution of foraging egrets and herons (Ardeidae) in the rice fields of
central Japan. Biological Conservation 83(2): 221–230; http://doi.org/10.1016/S0006-3207(97)00054-2
Ma, Z.,
Y. Cai, B. Li & J. Chen (2010). Managing wetland
habitats for waterbirds: an
international perspective. Wetlands 30(1): 15–27; http://doi.org/10.1007/s13157-009-0001-6
MacArthur, R.H. & J.W. MacArthur (1961). On bird species diversity. Ecology 42(3):
594–598; http://doi.org/10.2307/1932254
Maltby, E. & R.E. Turner (1983). Wetlands of the world. Geographical Magazine 55:
12–17.
Manral, U., A. Raha, R. Solanki, S.A. Hussain,
M.M. Babu, D. Mohan, G.G. Veeraswami, K. Sivakumar & G.
Talukdar (2013). Plant species of Okhla
Bird Sanctuary: a wetland of Upper Gangetic
Plains, India. Check List 9(2):
263–274; http://doi.org/10.15560/9.2.263
Morrison, M.L. (1986). Bird Populations as indicators of environmental
change, pp. 429–451. In:
Johnston, R. (ed.). Current Ornithology - Vol. 3. Springer, Boston, 522pp.
Mukherjee, A., C.K. Borad & B.M. Parasharya
(2002). A study of the ecological
requirements of waterfowl at man-made reservoirs in Kheda District,
Gujarat, India, with a view
towards conservation,
management and planning. Zoos’ Print Journal
17(5): 775–785; http://doi.org/10.11609/JoTT.ZPJ.17.5.775-85
Nudds, T.D. &
J.N. Bowlby (1984). Predator-prey size relationships in North American dabbling ducks.
Canadian Journal of Zoology 62(10): 2002–2008.
Panigrahy, S., T.V.R.
Murthy, J.G. Patel & T.S. Singh (2012). Wetlands of
India: inventory and assessment at 1:50,000 scale using geospatial techniques. Current Science 102(6): 852–856.
Prasad, S.N., T.V. Ramachandra, N. Ahalya, T. Sengupta, A. Kumar, A.K. Tiwari,
V.S. Vijayan & L. Vijayan
(2002). Conservation of wetlandsof India - a review.Tropical Ecology 43(1):
173–186.
Praveen, J., R. Jayapal
& A. Pittie (2016). A checklist of the birds of India. Indian Birds 11(5&6):
113–172.
Rahmani, A.R.
(1989). Status of the Black-necked Stork Ephippiorhynchus asiaticus
in the Indian subcontinent. Forktail 5: 99–110.
Reginald, L.J., C. Mahendran, S.S. Kumar
& P. Pramod (2007). Birds of Singanallur Lake, Coimbture, Tamilnadu. Zoos’ Print Journal22(12): 2944–2948; http://doi.org/10.11609/JoTT.ZPJ.1657.2944-8
Stewart, R.E., Jr. (2007).Technical Aspects of
wetlands: Wetlands as Bird Habitat.National Water Summary on Wetland Resources,
United States Geological Survey Water Supply paper 2425, 86pp.
Subramanya, S. (1996). Distribution,
status and conservation of Indian heronries. Journal
of the Bombay Natural History Society 93(3): 459–486.
Surana, R., B.R. Subba & K.P. Limbu (2007). Avian diversity during rehabilitation stage of Chimdi
Lake, Sunsari, Nepal. Our
Nature 5(1): 75–80; http://doi.org/10.3126/on.v5i1.802
Tak, P.C., J.P.
Sati & A.N. Rizvi (2010). Status of water birds at Hathnikund
Barrage wetland, Yamunanagar District, Haryana,
India. Journal of Threatened Taxa 2(4): 841–844; http://doi.org/10.11609/JoTT.o2200.841-4
Tews, J., U. Brose, V. Grimm, K. Tielbörger,
M.C. Wichmann, M. Schwager
& F. Jeltsch (2004). Animal
species diversity driven by habitat heterogeneity/diversity: the importance of
keystone structures. Journal of Biogeography 31(1): 79–92; http://doi.org/10.1046/j.0305-0270.2003.00994.x
Torell, M., A.M. Salamanca & M. Ahmed (2001). Management
of wetland resources in the Lower Mekong Basin: issues and future directions. Naga
24(3 &4): 4–10.
Torre-Cuadros,
M.D.L.A.L., S. Herrando-Perez
& K.R. Young (2007). Diversity and
structure patterns for tropical montane and premontane forests of central Peru, with an assessment of
the use of higher-taxon surrogacy. Biodiversity and Conservation
16(10): 2965–2988.
Urfi, A.J.
(2003). The birds of Okhla
Barrage Bird Sanctuary, Delhi, India. Forktail 19:
39–50.
Verma, A., S. Balachandran, N. Chaturvedi & V. Patil (2004).
A preliminary
report on the biodiversity
of Mahul Creek, Mumbai, India, with special reference to Avifauna. Zoos’Print
Journal 19(9): 1599–1605; http://doi.org/10.11609/JoTT.ZPJ.1172.1599-605
Wiens, J.A.
(1989). The Ecology of Bird Communities. Cambridge University Press, Cambridge, 539pp.
Zedler, J.B. & S. Kercher (2005). Wetland
resources: status, trends, ecosystem services, and restorability. Annual
Review of Environment and Resources 30: 39–74; http://doi.org/10.1146/annurev.energy.30.050504.144248