![]()
Journal of Threatened
Taxa | www.threatenedtaxa.org | 26 April 2018 | 10(5): 11583–11594

Comparative
cross-sectional survey on gastrointestinal parasites of captive, semi-captive,
and wild Elephants of Sri Lanka
Nirupama Abeysekara
1, R.P.V. Jayanthe Rajapkse
2 & R.S. Rajakaruna
3
1,3 Department of Zoology, University of Peradeniya, Peradeniya, 20400,
Sri Lanka
2 Department of Veterinary Pathobiology, Faculty
of Veterinary Medicine and Animal Sciences, University of Peradeniya,
Peradeniya 20400, Sri Lanka
1 niru163163@gmail.com,
2 jayanthar@pdn.ac.lk, 3 rupika.rajakaruna@yahoo.ca
(corresponding author)
Abstract:
Parasites can influence the fitness of
individuals particularly of small populations of endangered species. An island-wide, cross sectional, coprological
survey was carried out from 03 January to 30 October 2015, to determine the
gastrointestinal (GI) parasites of the Sri Lankan Elephant Elephas
maximus maximus. Fresh fecal
samples from wild, captive and semi-captive
elephants were collected and analyzed
using a modified salt floatation, Sheather’s sucrose
floatation, direct iodine smears, and sedimentation methods. Species
identification was done morphologically. Intensity of parasite infections was
determined using McMaster technique.
A total of 85 fecal samples (wild = 45,
semi-captive = 20, captive = 20) were analysed; 58 (68.2%) samples were
positive for GI parasites. Overall,
helminth infections (60.0%) were more common than
protozoan (37.6%) infections (Chi square test, χ2 = 8.499; p
< 0.001). In the captive elephants, however, more protozoan infections were
observed than helminthes, which could be due to
anthelminthic treatment. A
significantly higher prevalence of infection was observed in the wild elephants
(93.3%) compared to semi-captive elephants (55.0%; χ2 = 13.516;
p < 0.001) and captive elephants (25.0%; χ2 =32.289; p
< 0.001) but there was no significant difference in
the prevalence between captive and semi-captive elephants (χ2
=3.750; p = 0.053). Ten types of GI
parasites were observed, nine of which were recorded in wild elephants. Among them the most common infection was
strongyles (34.1%) with high intensity (440.1±295.2
EPG). Semi-captive elephants harbored five types of GI parasites, while captive
elephants had only three types. One
captive elephant at the Temple of the Tooth was infected with the tapeworm Anoplocephala sp. at low intensity of 50 EPG. Some of the GI parasites recorded are
highly pathogenic while others are incidental.
Keywords:
Gastrointestinal parasites, Elephants, Sri Lanka.
doi: http://doi.org/10.11609/jott.3406.10.5.11583-11594 | ZooBank:
urn:lsid:zoobank.org:pub:64AF5FAE-1424-42E0-B66A-6806925F945D
Editor:
Heidi S. Riddle,
Riddle’s Elephant and Wildlife Sanctuary, Arkansas, USA. Date
of publication: 26 April 2018 (online & print)
Manuscript
details: Ms # 3406 | Received 10 March 2017 | Final received 30
March 2018 | Finally accepted 04 April 2018
Citation: Abeysekara, N., R.P.V.J. Rajapkse & R.S. Rajakaruna (2018). Comparative cross-sectional survey on gastrointestinal
parasites of captive, semi-captive, and wild elephants of Sri Lanka. Journal of Threatened
Taxa 10(5): 11583–11594; http://doi.org/10.11609/jott.3406.10.5.11583-11594
Copyright: © Abeysekara et al. 2018. Creative Commons Attribution 4.0
International License. JoTT allows
unrestricted use of this article in any medium, reproduction and distribution
by providing adequate credit to the authors and the source of publication.
Funding: None.
Competing interests: The
authors declare no competing interests.
Author Details: Nirupama Abeysekara: completed her B.Sc. (Hons.)
in Zoology at the University of Peradeniya, Sri
Lanka. She is Following her M.Sc. in Molecular and
Applied Microbiology and a trainee at Veterinary Investigation and Research
Center, Peradeniya, Sri Lanka and also volunteering
as a Research Assistant at the Department of Zoology, University of Peradeniya. Rupika Subashini Rajakaruna: received her B.Sc. (Hons.)
and MPhil degrees in Zoology from the University of Peradeniya
and completed her Ph.D. at the Memorial University of Newfoundland, Canada. She
has over 20 years of teaching and research experience and currently holds the
Chair of Professor of Applied Zoology at the University of Peradeniya. R.P.V.
Jayantha Rajapakse:
graduated from the Faculty of Veterinary Medicine and Animal Sciences at the
University of Peradeniya, Sri Lanka. He is currently
a Senior Professor of Veterinary Parasitology at the Department of Veterinary Pathobiology,
Faculty of Veterinary Medicine and Animal Sciences, University of Peradeniya.
Author Contribution: NA: Collected and analysed the
fecal samples, Analysed data, wrote the manuscript;
RPVJR: Supervised the parasite egg identification; RSR: Designed the study,
supervised and edited the manuscript.
Acknowledgements:
Authors thank Shanmugasundaram Wijeyamohan for
his support in elephant dung collection.
INTRODUCTION
Globally, the Asian
Elephant is listed as ‘Endangered’ in the IUCN Red List of Threatened Species
(IUCN 2008) and is protected under the Convention on International
Trade in Endangered Species (CITES)
Act. Three subspecies of the Asian
Elephant are currently recognized: the Sri Lankan Elephant Elephas
maximus maximus, the
Indian Elephant Elephas maximus
indicus from the Asia mainland, and the Sumatran
Elephant Elephas maximus
sumatranus from the island of Sumatra in
Indonesia (IUCN 2017). Among these,
the Sri Lankan elephant has the highest genetic diversity (Fernando 2000;
Fleischer et al. 2001). Sri Lanka
has the highest density of elephants with over 10% of the global Asian Elephant
population in less than 2% of elephant range (Leimgruber
et al. 2003). In Sri Lanka, as in
the rest of Asia, the elephant has been closely associated with humans and has
played a central role in the country’s economy, conflicts, religion, and
culture for many millennia (Jayewardene 1994). It also holds an important position in
the religious and cultural traditions of the country and plays a significant
and high profile role in the country’s conservation efforts (Jayewardene 1994).
Elephants in the
wild are susceptible to many gastrointestinal (GI) parasites (Watve 1995; Dharmarajan 1999; Vidya & Sukumar 2002); in
captivity they have enhanced susceptibility as they are often confined to small
enclosures and/or maintained in damp unhygienic conditions (Chandrashekaran
et al. 1995). Strongyles
have been observed in African (Scott & Dobson 1985) and Asian wild
elephants as the most common GI parasite (Vidya &
Sukumar 2002).
In the Asian elephant the helminthes, such as Parabronema, Brumptia,
Pseudodiscus, Paramphistomum,
and Fasciola and Anoplocephala,
have been recorded (Bhalerao 1933; Gupta 1974; Chandrashekaran et al. 2009). The most common protozoans recorded are Entamoeba and coccidian cysts. Injuries,
parasitism, and gastrointestinal disease were reported as the most common
syndromes responsible for elephant morbidity (Miller et al. 2015). In Kenya, necropsy of 11 fresh wild
elephant carcasses revealed the death of the elephants were due to pathological
lesions on the intestinal mucosa and haemorrhages which were linked to
gastrointestinal parasitism (Obanda et al. 2011).
Studies carried out
on Sri Lankan elephant GI parasites record many nematodes: Murshidia murshidia, M. falcifera, M. longicaudata, Quilonia renniei, Equinubria sipunculiformis, Decrusia additictia (Seneviratna
1955; Fernando & Fernando 1961; Fowler & Mikota
2008), Grammocephalus hybridatus,
Parabromina smithi
(Seneviratna & Jayasinghe
1968; Perera et al. 2014), Q. travancra,
Choniangium epistomum, Amira pileata, Bathmostpmum sangeri
(Kuruwita & Vasanthathilake
1993), and trematodes: Cabboldia
elephantis (Seneviratna
& Jayasinghe 1968), liver fluke Fasciola
jacksoni (Perera & Rajapakse 2009), and schistosome,
Bivitellobilharzia nairi
(Agatsuma et al. 2004). More recent studies report strongyle infections in captive and wild elephants in Sri
Lanka (Heinrich 2016; Abeysinghe et al. 2017), and
unidentified protozoan cysts in captive elephants (Aviruppola
et al. 2016). Except for the
studies carried out in the 1950s and 1960s (Seneviratna 1955; Fernando & Fernando 1961; Seneviratna & Jayasinghe
1968), recent reports on elephant parasites are focused either on a specific
parasite or a particular group of hosts. Here we carried out an island-wide,
comparative, coprological survey of GI parasites
collecting samples from the wild, captive, and semi-captive elephants of Sri
Lanka.
MATERIALS AND
METHODS
An island-wide
collection of fecal samples from wild, captive, and
semi-captive elephants was carried out from 03 January to 30 October 2015. According to the most recent island-wide
survey of wild elephants in Sri Lanka carried out in 2011, a minimum of 5,879
animals was estimated (Dissanayake et al. 2012; Santiapillai & Wijeyamohan
2013). There are about 150 captive
elephants and 45 semi-captive elephants in Sri Lanka (Fernando et al. 2011).
Wild Elephants
The wild elephants
once found throughout Sri Lanka are restricted mainly to the lowland dry zone,
which is approximately 60% of the island.
Wild elephants are found in protected areas such as national parks
including Wilpattu, Yala, Udawalawe, Maduru Oya, Minneriya, Wasgamuwa and Lunugamwehera. Some are not limited to protected areas
and are found outside where food and water is plentiful. Wild elephants are free grazers or
browsers and do not receive any veterinary care.
Captive Elephants
Elephants that are
kept permanently under human control are known as captive elephants. In Sri Lanka, captive
elephants are kept by temples, private owners, Pinnawala
Elephant Orphanage (PEO), Millenium Elephant
Foundation (MEF) in Kegalle, and National Zoological
Gardens at Dehiwala. In addition, at the Temple of the Tooth
in Kandy there are 12 elephants, all of which are males and consisting of two tuskers from India, two tuskers
from Myanmar, one tusker from Thailand, and four tuskers and three non-tuskers
from Sri Lanka. These elephants do
not interact with wild ones. They
have a great cultural and economic importance (Fernando et al. 2011). Elephants are used in religious
festivals and parades; in the past they were used in the transport of
timber. Elephants are of great
economic value mainly for tourism.
The MEF is a private enterprise, which serves as a retirement home for
working elephants. Currently there
are 10 elephants at MEF; the National Zoo in Dehiwala
has eight elephants, while there are about 93 elephants in PEO. Veterinary care for captive elephants is
provided and they are dewormed at least twice a year. Febantel
(Rintal®) is a first line drug for deworming
elephants and Mebendazole is also commonly used.
Semi-captive Elephants
The Department of
Wildlife Conservation (DWC), Sri Lanka, has established the Elephant Transit
Home (ETH) in Udawalawe where semi-captive elephants
are kept (Santiapillai & Sukumar
2006). The aim is to conserve
elephants outside the protected areas.
Mostly, wild baby elephants that are orphaned due to the death of the
mother or abandonment are brought to the ETH which takes care of them until
they are fit enough to be released back to the wild, usually after 5–6
years. They do not interact with
wild elephants while they are at the ETH.
These elephants receive regular veterinary care (e.g., deworming,
vaccinations), a standard diet, and bathing. Currently, there are 45 elephants in the
ETH. They remain as a group during
the day and are kept in a stall at night.
All the elephants are bottle fed seven times throughout the day.
Study sites
Fecal samples of wild elephants were collected
from four national parks: Minneriya, Udawalawe, Maduru Oya, Yala, and from Mannar District closer to Wilpattu
National Park. All the samples from
semi-captive elephants were collected from the ETH in Udawalawa. Fecal samples
from captive elephants were collected from the elephants in the Temple of the
Tooth in Kandy, MEF in Kegalle, and from a private
elephant owner in Biyagama (Fig. 1).
Udawalawe National Park contains 400 elephants and
lies on the boundary of Sabaragamuwa and Uva provinces in the Wet Zone of Sri Lanka. The park has an annual rainfall of
1,500mm, most of which falls during the months of October to January and March
to May. The average annual
temperature is about 27–28 0C, while relative humidity
varies from 70% to 82%. This
national park spans approximately 31,000ha. Yala National
Park is the most visited and second largest national park in Sri Lanka with
97,880ha and 350 elephants. It is
located in Uva province with an elevation of
64m. It is situated in dry
semi-arid climatic region and rain is received mainly during the northeast
monsoon. The mean annual rainfall
ranges between 500–775 mm while the mean temperature ranges between 26.40C
in January to 300C in April.
Maduru Oya National
Park is located in Eastern and Uva provinces in the
dry zone with about 200 elephants.
Altitude ranges from 20–60 m.
The northeast monsoon is instrumental in the mean annual rainfall of
1,650mm, and the mean annual temperature is 270C. Maduru Oya National Park is spread over 58,850ha with a special
feature of an 8km long rocky range of hills to the southwest of the park. Mannar
District is closer to Villpatthu National Park and is
one of the 25 districts of Sri Lanka with 290C annual
temperature. The mean annual
rainfall ranges between 550–580 mm whereas the area only experiences the
tail end of the northeastern monsoon (Department of
Wildlife Conservation 2015).
Sample collection
About 50g of fresh fecal samples were collected in a zip-locked plastic bag by
inverting the bag and scooping the needed amount into the bag, and then samples
were stored in a cooler. The
collection was done soon after defecation whenever possible and each site was visited only once and each elephant was sampled once.
Samples were labeled and brought to the laboratory
and were stored at 40C until processed. Information on the study animals was
recorded using a questionnaire.
Sample analysis
Fecal samples were analyzed
in the parasitology laboratories of the Department of Veterinary Pathobiology
in the Faculty of Veterinary Medicine and Animal Science at the University of Peradeniya.
Qualitative and quantitative analysis were carried out to determine the
types of gastrointestinal parasites (GI) and their intensities. Under qualitative analysis, direct
saline and iodine mounts, modified salt flotation, Sheather’s
modified sucrose flotation method, and sedimentation techniques were carried
out for each of the samples simultaneously (WHO 1991). Intensity of infection in the positive
samples was calculated using McMaster counting technique; number of eggs or
cysts/oocysts per gram (EPG/CPG/OPG) of feces was calculated.
Modified salt floatation method
About 50g of feces was measured and transferred into a 50ml capped
centrifuge tube with 45ml distilled water. Feces were
mixed thoroughly using a wooden applicator to get a clear solution. This suspension was centrifuged at
2,016g for 20min. The supernatant
was removed using a suction pump.
Again 45ml of distilled water was added and it was centrifuged for 20min
at 2,016g. The supernatant was
discarded. Once the supernatant was
removed, 45ml of salt solution was added in to the pellet in the butt of the
tube. This was centrifuged at
2,016g for 20min. Approximately,
5ml of the supernatant with floating parasitic eggs was removed in to a 15ml
centrifuge tube. The total volume
was made up to 15ml by adding distilled water and the tube was centrifuged at
2,016g for 10min at 160C.
Supernatant was removed and pipetted in to a 1.5ml Eppendorf®
microfuge tube using a Pasteur pipette.
Distilled water was added to make it up to 1.5ml and the tube was
centrifuged for 10min at 2,744g at 160C in the micro
centrifuge. The supernatant was
decanted and the pellet was mixed thoroughly with 0.5ml of the
supernatant. Microscope slides were
prepared using about 0.1ml of the suspension and covered with a cover slip
without staining. Three smears were
observed from each sample under ×10 and ×40 objectives (WHO 1991).
Different stages of
parasites (eggs, larvae, cysts) were morphologically identified with the aid of
experts, standard keys, and literature (Soulsby 1982;
Fowler & Mikota 2008). All stages were photographed and
measurements were taken using Primostar Zeiss trinocular microscope.
Statistical analysis
The prevalence of
parasitic infection in wild, semi-captive, and captive elephants was calculated
using the following formula: Prevalence = (Infected number ÷ Individuals examined) x 100%. The prevalence of infections in wild,
semi-captive, and captive elephants and the difference in helminth
and protozoan infections were compared using a Chi squared test.
RESULTS
A total of 85 fecal samples were collected: 45 from wild elephants, 20
from semi captive elephants, and 20 from elephants in captivity.
Prevalence of GI parasites
Of the samples
collected 58 (68.2%) were infected with one or more GI parasites of nematodes, trematodes, cestodes, and
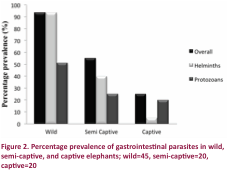
protozoans. Overall, helminth infections (60.0%) were significantly more common
compared to the protozoan (37.6%) infections (Chi square test, χ2
= 8.499, p < 0.001; Fig. 2).
Among the three host categories, the wild elephants had the highest
prevalence (93.3%) of GI infections where 42 out of 45 individuals were
infected. They all had helminth infections (93.3%) which were significantly higher
than the protozoa infections (51.1%; χ2 = 19.994, p <
0.001). All the wild elephants
sampled from Minneriya National Park, Udawalawe National Park, and from Mannar
District were infected with GI parasites (Table 1). The second highest prevalence of GI
infections was recorded from semi-captive elephants (55.0%) where 11 elephants
were positive. There was no
difference between the helminths (40.0 %) and
protozoan (25.0 %) infections (χ2 = 1.026, p =
0.311). When the prevalence of GI
parasites in the two groups was compared, a significantly higher prevalence of
infection was observed in the wild elephants compared to the semi-captive
elephants (χ2 = 13.516, p < 0.001). The captive elephants had the lowest GI
infections (25.0%) among the three groups where only five elephants out of 20
were positive for GI parasites.
Protozoan infections were more common (20.0 %) in captive elephants than
helminth (5.0%) infections, but this difference was
not statistically significant (χ2 =2.057; p =
0.151). Among the captive
elephants, only one elephant sampled from Kandy (14.3%) had a helminth infection only (Table 1). There was a highly significant
difference of parasitic prevalence between captive and wild elephants (χ2
= 32.289; p < 0.001) but not between the captive and the semi-captive
elephants (χ2 = 3.750; p = 0.053).
Table 1. Prevalence of helminths and protozoans in the fecal
samples of captive, semi-captive, and wild elephants collected from different
sites in Sri Lanka
|
Collection site (n) |
Prevalence (%) |
|||
|
Overall |
Helminths |
Protozoans |
||
|
Wild |
Yala NP (8) |
87.5 |
87.5 |
62.5 |
|
Maduru Oya NP (10) |
80.0 |
80.0 |
50.0 |
|
|
Minneriya NP (10) |
100 |
100 |
40.0 |
|
|
Udawalawe NP (10) |
100 |
100 |
50.0 |
|
|
Mannar (7) |
100 |
100 |
57.1 |
|
|
Semi- Captive |
Udawalawe ETH (20) |
55.0 |
40.0 |
25.0 |
|
Captive |
Kandy - TTR (7) |
28.6 |
14.3 |
14.3 |
|
Kegalle - MEF (10) |
20.0 |
0.0 |
20.0 |
|
|
Biyagama (3) |
33.3 |
0.0 |
33.3 |
|
|
|
Total (85) |
68.2 |
60.0 |
37.6 |
NP = National Park; TTR = Temple of Tooth Relic; ETH=
Elephant Transit Home; MEF= Millennium Elephant Foundation
Types of GI parasites
A total of 10
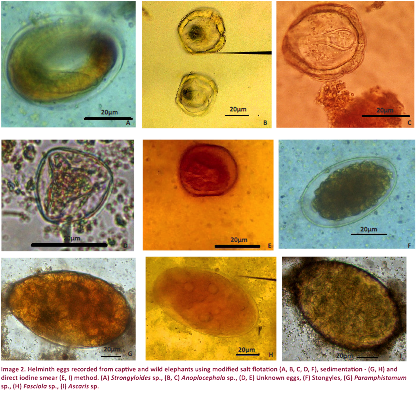
parasite species were found in the elephants (Table 2). Among them the most common helminth infection was strongyles
(34.1%) followed by Ascaris sp. (22.9%). Among the protozoans, Entamoeba sp. (18.8%) was the most prevalent
followed by coccidia cysts (15.3%). Wild elephants harbored
more parasite species than the other two groups. Overall nine types: strongyles,
Strongyloides sp., Ascaris
sp., Fasciola sp., Paramphistomum
sp., unidentified helminth eggs, Entamoeba
sp., coccidian cysts, and unidentified protozoan cysts were recorded (Table
2). In semi-captive and captive
elephants five types and three types of parasites were recorded,
respectively. The wild elephants
had all the parasites recorded in the present study except Anoplocephala
sp., which was recorded only from one captive elephant from the Temple of
the Tooth in Kandy. All the samples
from Minneriya National Park were positive for strongyles. All
three elephant groups had Entamoeba sp.
infections. Among helminths, strongyles, Ascaris sp., and Fasciola
sp. were recorded from both semi-captive and wild elephant categories (Table
2). Unidentified protozoan cysts
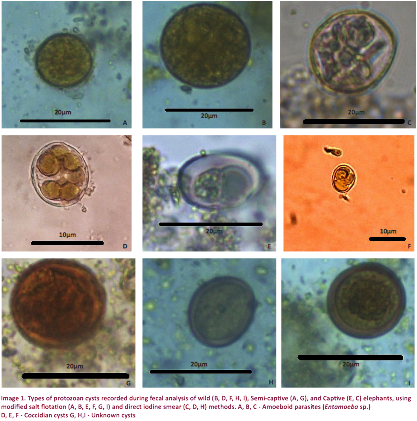
were found in semi-captive and wild elephants (Image 1). The captive elephants harbored three parasite species (Anoplocephala
sp., Entamoeba sp., and coccidia
cysts; Table 2). None of the
elephants in MEF and from private owners in Biyagama
had GI helminths. Four samples were positive (20.0%)
for protozoans (Entamoeba sp., coccidia cysts).
Entamoeba sp. was found only in two
elephants from MEF in Kegalle. None of the other captive elephants were
positive for Entamoeba infection. Coccidia cysts
were positive in 1 sample from Temple of the Tooth and in 1 sample from Biyagama.
Mixed infections
Mixed infections
were more common (47.1%) than single infections (21.2%). The percentages of elephants having
mixed infections of 2, 3, and 4 parasites were 24.7%, 17.6%, and 4.7%,
respectively. Multiple infections
of helminthes (30.6%) were more common than those of
protozoans, which were found only in three elephants (3.5%). Multiple helminth
infections were not recorded in semi-captive and captive elephants. Twenty-three wild elephants were
infected with both helminth and protozoan
infections. Eight semi-captive
elephants were infected with single GI infection (40.0%) and three were
infected with mixed infections (15.0%).
Among the eight single infections, six samples were positive for helminths and two samples were positive for protozoans.
Intensity of GI parasites
A high intensity of
infection was recorded for strongyles in wild
elephants (Table 3). The highest helminth egg count of 1,100 EPG of strongyles
was recorded in a wild elephant from Minneriya
National Park. In general, all the
infections in wild elephants had a higher intensity than those in the captive
and semi-captive animals. Highest
number of cysts (340 OPG) of Entamoeba sp. was
recorded from a wild elephant in Maduru Oya National Park.
The intensity of all the infections in the wild elephants was higher
compared to semi-captive and captive elephants. The intensity of common helminth and protozoans infections was lower in captive and
semi-captive elephants than in wild elephants. Unknown cysts were found in semi-captive
and wild elephants; among them the lowest cyst count (50 OPG) was recorded from
a semi-captive elephant in ETH (Image 1). Anoplocephala
sp. was recorded at low EPG (40) in only one captive elephant at the Temple of
the Tooth in Kandy. Coccidia cysts were found in wild and captive elephants;
among them the lowest OPG (45) was recorded from a captive elephant in Biyagama (Image 2).
Table 2. Types of
parasites and their prevalence in wild, semi-captive, and captive elephants in
Sri Lanka (n =85)
|
Parasitic group |
Parasite species |
Prevalence of infection (%) |
|||
|
Overall |
Captive |
Semi captive |
Wild |
||
|
Helminths |
Strongyles |
34.1 |
- |
20.0 |
55.6 |
|
|
Strongyloides sp. |
16.5 |
- |
- |
31.1 |
|
|
Ascaris sp. |
22.9 |
- |
10.0 |
37.8 |
|
|
Anoplocephala sp. |
1.2 |
5.0 |
- |
- |
|
|
Paramphistomum sp. |
3.5 |
- |
- |
6.7 |
|
|
Fasciola sp. |
12.9 |
- |
10.0 |
20.0 |
|
|
Unknown eggs |
5.9 |
- |
- |
11.1 |
|
Protozoans |
Entamoeba sp. |
18.8 |
10.0 |
20.0 |
22.2 |
|
|
Coccidia cysts |
15.3 |
10.0 |
- |
24.4 |
|
|
Unknown cysts |
5.9 |
- |
10.0 |
6.7 |
|
|
Total |
68.2 |
25.0 |
55.0 |
93.3 |
Table 3. Mean Eggs per gram (EPG) or Oocysts per gram (OPG) counts of different parasites in the
captive, semi-captive, and wild elephants
|
Parasite species |
Mean EPG or OPG counts (±SD) |
|||
|
Captive |
Semi-captive |
Wild |
||
|
Eggs |
Strongyles |
- |
50.0±9.1 |
440.1±295.2 |
|
|
Strongyloides sp. |
- |
- |
225.8±157.7 |
|
|
Ascaris sp. |
- |
95.0±7.1 |
251.2±225.1 |
|
|
Anoplocephala sp. |
40 |
- |
- |
|
|
Paramphistomum sp. |
- |
- |
123.3±94.5 |
|
|
Fasciola sp. |
- |
60.0±14.1 |
152.2±97.7 |
|
|
Unknown eggs |
- |
- |
82.5±34.9 |
|
Cysts |
Entamoeba sp. |
75.0±35.4 |
58.0±22.2 |
169.8±125.4 |
|
|
Coccidia cysts |
55.0±7.1 |
- |
92.7±52.1 |
|
|
Unknown cysts |
- |
30.0±14.1 |
65.0±35.4 |
*Note -SD = Standard Deviation.
Table 4. Percentage prevalence of single
and mixed infections of gastrointestinal parasitic groups in wild,
semi-captive, and captive elephants in Sri Lanka
|
Parastic group |
Infection Type |
Prevalence of infection (%) |
|||
|
Overall |
Captive |
Semi captive |
Wild |
||
|
Helminths |
Single |
29.4 |
5.0 |
40.0 |
35.6 |
|
Mixed |
30.6 |
- |
57.8 |
- |
|
|
Protozoans |
Single |
34.1 |
20.0 |
20.0 |
46.7 |
|
Mixed |
3.5 |
5.0 |
4.4 |
- |
|
DISCUSSION
The present study
reports an overall prevalence of 68.2% of GI parasites in elephants of Sri
Lanka; the infection in wild elephants (93.3%) was significantly higher than
that of the captive (55.0%) and semi-captive elephants (25.0%). Unlike the wild elephants, the captive
and semi-captive elephants receive regular deworming which is the main reason
for having a low prevalence of GI parasites in these two groups. In general, oral deworming treatments
are given two to three times per year with special treatments if elephants show
clinical symptoms. Febantel and Mebendazole
are the commonly used anthelminthics for both captive
and semi-captive elephants in Sri Lanka (personal communication with the
veterinarian). Febantel
is a broad spectrum anthelminthic used against GI parasites including Giardia,
roundworms, hookworms, whipworm, ascarids, and
tapeworms (Tiwari & Rao
1996). Deworming of both adult
elephants and calves is done with Febantel twice a
year @ 5–10 mg/kg body weight.
Mebendazole is a highly effective,
broad-spectrum anthelmintic for the treatment of nematode infestations,
including roundworm, hookworm, whipworm, threadworm, and the intestinal form of
trichinosis prior to its spread into the tissues beyond the digestive tract (Tiwari & Rao 1996). The prevalence of helminth
infections was higher in wild elephants than the other two groups. This could be because the anthelminthic
drugs mainly target the helminths. Irrespective of
regular deworming treatments, one third of Biyagama
captive elephants were infected.
This could be attributed to the method of deworming where individuals
were treated separately, while the other captive elephants were treated
simultaneously. Moreover, sampling of elephants in Biyagama
was done five months after the last deworming while sampling of other two sites
was carried out within 2 to 3 months after deworming. Furthermore, in Biyagama
the space limitation for the captive animals can aggravate the prevalence of GI
parasites. All the wild elephants
sampled from Minneriya and Udawalawe
National Parks were infected. Due
to the water scarcity in these national parks, many elephants congregate and
depend on a single water hole. This
tends to increase the contamination rate as they defecate on the ground and
there is a higher possibility of infection of the whole herd when one
individual is infected. Potential
factors determining the transmission of GI parasites in the wild include
environmental conditions that affect the viability and behaviour of parasite propagules, as well as feeding, movement, and defecation
patterns of the host, which determine the parasites encountered (Watve 1995; Vidya & Sukumar 2002).
A total of 10
species of GI parasites was found in the elephants. All the helminth
species except Anoplocephala were recorded in
wild elephants. None of the captive
elephants had any helminths except Anaplocephala.
Semi-captive elephants had strongyles, Ascaris, and Faciola
infections. All three protozoans
recorded were found in wild elephants.
Entamoeba was reported both in captive
and semi-captive elephants. Strongyles was
the most common infection. The most
common helminth was strongyle
followed by Ascaris. A recent study carried out in Sri Lanka
at Pinnawela Elephant Orphanage (PEO), Galgamuwa wild elephants, and with some privately owned
elephants by Abeysinghe et al. (2017) reported that
100% of wild elephants and 90% of privately owned captive elephants were
infected with strongyles, while only 38% of elephants
in PEO harbored that infection. Vanitha et al.
(2011) reported the prevalence of strongyles as 37%
in captive elephants in Tamil Nadu, India.
They show that the prevalence varies significantly across seasons, with
the highest rate during summer (49%) followed by monsoon (41%) and the lowest
rate during winter (15%).
Furthermore, they have shown that male elephants have a lower parasite
prevalence compared to females, and the age classes show no difference. Previous studies from India also record strongyles as the most comment type of GI parasite (Watve 1995; Chandrasekharan et
al. 1995; Vidya & Sukumar
2002; Chandrasekharan et al. 2009). Saseendran et
al. (2003) reported 10% of captive elephants had strongyles
in Kerala, India. There is a
correlation between the pathogenecity of the parasite
and the number of eggs that they produce.
In the present study, the strongyles were not
found in captive elephants. Anthelminthics against strongylosis
in captive elephants are effective (Chandrasekharan
1992; Suresh et al. 2001).
Ascaris infections were recorded in wild and
semi-captive elephants. Ascaris sp. is known
to evolve anthelminthic resistance, which could be a possible reason for the
presence of the parasite in semi-captive elephants, even with regular
anthelmintic treatments. It may
also be due to treating with broad-scale anthelminthics
at sub-curative dosages, which eventually leads to the development of
resistance. Resistance has been
recorded in both Asian and African Elephants in India (Bapu
1936), Nigeria (Mbaya et al. 2012), and Bangladesh (Rahman et al. 2014).
The other helminth infections recorded were Strongyloides sp., Paramphistomum
sp., Fasciola sp. These have also been reported in
elephants in India (Varadharajan & Kandasamy 2000) and in Borneo (Hing
et al. 2013).
One captive elephant
was infected with the tapeworm Anoplocephala
sp. at low intensity of 40 EPG.
This elephant was the oldest male elephant belonging to the Temple of
the Tooth and was in musth condition at the time of
sampling, which was done five months after a deworming treatment. Although all the elephants were kept
together, none of the six other elephants sampled from the same temple had the
infection. The oribatid
mite acts as the intermediate host transmitting the infection and can easily
spread the infection to other individuals in the vicinity. These six samples however, were
collected two months after the last deworming, which may be a reason for the
absence of Anoplocephala infection
among them. Moreover, cestode eggs are not equally distributed in the fecal matter, probably due to the eggs being shed as gravid
proglottids detached from tapeworm. Some studies report that Anoplocephala eggs are difficult to find in fecal samples (Ihler et al. 1995;
Nilsson et al. 1995) and suggest using modified methods and 30–40 g of feces.
Perera et al. (2014) recorded adult tapeworm Anoplocephala sp. from wild elephants in the Udawalawe National Park and the identifications were done
using morphometric features and molecular characterization of its 2s and 28s
genes. Anoplocephala
has been found in both Asian and African captive elephants where Obanda et al. (2011) reported that Anoplocephala
manubriata was common among wild African
Elephants. It has been recorded in
the Asian elephant in Kerala, India (Warren 1996), and in Tamil Nadu state in Mudumalai, Anamalai, and Sathyamangalam forests in India (Nishanth
et al. 2012).
All protozoan
parasites recorded in the present study (Entamoeba
sp., Coccidian cysts, and unknown cysts) have been previously
recorded in elephants in South Africa (Eloff &
van Hoven 1980; Samuel 2001).
Protozoans including coccidian parasites such as Eimeria
sp., Isospora sp. (Mbaya 2013), are commonly found in a wide variety of
species but rarely cause disease in free-living elephants (Samuel 2001). In addition, GI protozoans like Cryptosporidium,
Cyclospora, and Giardia have been
reported in captive African and Asian Elephants (Majewska
et al. 1997) but were not found in the present study. There are some non-pathogenic protozoans
that can be found in elephants, including in the amoeboid group (Begon 1995).
They do not harm elephants, even those with weak immune systems. Symptomatic elephants that are found to
have these protozoa in their feces should be examined
for other causes of their symptoms.
All the wild
elephants had mixed infections. A higher frequency of mixed infections in wild
elephants could be due to animal movement and grazing behavior. When there is greater freedom of animal
movement, it can result in feeding at a greater variety of locations and on
more different types of fodder thus increasing exposure to a greater variety of
endoparasites (Nunn et al. 2003). Moreover, the presence of one parasite
species may facilitate the presence of the other species (Fontanarrosa
et al. 2006) since the elephants that have mixed infections with higher
parasite intensities are known to have less immunity. Mixed infections of strongyles
and Strongyloides sp. are common in
wild elephants (Watve 1995; Dharmarajan
1999; Nishanth et al. 2012).
All
gastrointestinal parasites are not equal, some are highly pathogenic and some
are incidental. The presence of
parasites in the fecal samples of elephants does not
necessarily mean they are sick, or will be sick, nor does it mean that the
animal should be treated. Gaur et
al. (1979) stated that wild animals in a free-living state are generally
infected with numerous parasites, but these cause little harm to them unless
they are physiologically or nutritionally stressed. Understanding the infections in wild
animals is important since infections could result in die-offs of elephants
during extreme stress conditions.
In 1995, three Sumatran Elephants (Elephas
maximus sumatranus)
died suddenly of helminth infection in the Way Kambas National Park, Indonesia. Postmortem
examination revealed that the GI tracts of all three animals were infected with
Murshidia falcifera
(Nematoda), Hawkesius
hawkesi, Pfenderius papillatus (Digenea), and Cobboldia elephantis
(Diptera) (Matsuo et al. 1998).
The baseline
information of disease prevalence of already threatened taxa is important in
understanding the role of disease in provoking endangerment. Although there are some studies on GI
parasites of elephants in Sri Lanka, most of them were focused mainly on one
type of elephant group. This study,
however, provides a comprehensive survey of wild, captive, and semi-captive
elephants in Sri Lanka. All the
wild elephants sampled from Minneriya National Park, Udawalawe National Park and from Mannar
District were infected with GI parasites and some with very high
intensities. Deaths due to
parasitism have been reported from elsewhere. Necropsy of 11 fresh wild African
Elephant carcasses in Kenya revealed pathological lesions on the intestinal
mucosa and haemorrhages, which were linked to parasitism (Obanda
et al. 2011). It is likely that
starvation and dehydration could have triggered a vicious cycle of host
malnourishment, a result of combined inadequate food and nutritional
deprivation by intestinal parasites, which lead to emaciation, pathology, and
death (Obanda et al. 2011). So it is important to monitor
mortalities of wild elephants and carry out post-mortem sampling to determine
whether the cause of death was due to GI infections. Estimation of GI parasitic egg burdens
is also key for designing appropriate treatment or
management regimes in captive host populations. Therefore it is vital to do a fecal egg count prior to deworming semi-captive and captive
elephants. As stated by Miller et
al. (2015), there is a need to identify strategic investments in Asian Elephant
health that will yield maximal benefits for overall elephant health and conservation as prevention is often the most cost-effective
approach.
References
Abeysinghe, K.S., A.N.F. Perera,
J. Pastorini, K. Isler, C. Mammides & P. Fernando (2017). Gastrointestinal strongyle infections in captive and wild elephants in Sri
Lanka. Gajah 46: 21–27.
Agatsuma, T., R.P.V.J. Rajapakse, V.Y. Kuruwita, M. Iwagami & R.C. Rajapakse
(2004). Molecular
taxonomic position of the elephant schistosome, Bivitellobilharzia nairi,
newly discovered in Sri Lanka. Parasitology
International 53(1): 69–75.
Aviruppola, A.J.M., R.P.V. Rajapakse
& R. Rajakaruna (2016). Coprological survey of gastrointestinal parasites of mammals in Dehiwala National Zoological Gardens, Sri Lanka. Ceylon
Journal of Science 45(1): 83–96; http://doi.org/10.4038/cjs.v45i1.7367
Bapu, S. (1936). A short note on
elephants and a few of their common diseases. Indian
Veterinary Journal 13(1): 36–43.
Begon, M. & R.G. Bowers
(1995). Beyond host-pathogen
dynamics, pp. 478–509.
In: Grenfell, B.T. & A.P. Dobson (eds.). Ecology of
Infectious Diseases in Natural Populations. Cambridge
University Press, Cambridge.
Bhalerao, G.D. (1933). The trematode
parasites of the Indian Elephant, Elephas indicus. Indian Journal of
Veterinary Science and Animal Husbandry 3: 103–115.
Chandrashekaran, K. (1992). Prevalence of infectious diseases
in elephants in Kerala and their treatment. In: Silas, E.G., M.K. Nair
& G. Nirmalan (eds.). The Asian Elephant:
Ecology, Biology, Diseases, Conservation and Management. Kerala
Agricultural University, India.
Chandrasekharan, K., K. Radhakrishnan,
J.V. Cheeran, K.N. Muraleedharan
& T. Prabhakaran (1995). Review of the
incidence, etiology and control of common diseases of
Asian elephants with special reference to Kerala, pp. 439–449. In:
Daniel J.C. & H.S. Datye (eds.). A Week with Elephants. International
Seminar on the Conservation of Elephant, India.
Chandrasekharan, K., K. Radhakrishnan,
J.V. Cheeran, K.N. Muraleedharan
& T. Prabhakaran (2009). Review of the
incidence, etiology and control of common diseases of
Asian elephants with special reference to Kerala, pp. 92–100. In: Ajitkumar G., K.S. Anil & P.C. Alex (eds.). Healthcare Management of Captive Asian Elephants. Kerala
Agriculture University, India.
Department of Wildlife Conservation
(2015). Ecosystem conservation
and management. Electronic version,
accessed on 10 September 2015.
Dharmarajan, G. (1999). Epidemiology of helminth
parasites in wild and domestic herbivores at the Mudumalai
Wildlife Sanctuary, Tamil Nadu. M.V.Sc Thesis.
Tamil Nadu Veterinary and Animal Sciences University,
Chennai.
Dissanayake, S.R.B., R. Marasinghe,
M. Amararathne, S. Wijeyamohan,
P. Wijeyakoon & C. Santiapillai
(2012). The First National
Survey of Elephants in Sri Lanka, p. 113. A
Report Prepared for The Department of Wildlife Conservation. Center for the
Study of Asian Elephant at Rajarata University of Sri
Lanka, Mihintale, Sri Lanka.
Eloff, A.K. & W. van Hoven (1980). Intestinal protozoa of
the African Elephant Loxodonta africana
(Blumenbach). South African Journal of Zoology
15(2): 83–90.
Fernando, P. (2000). Elephants in Sri Lanka: past, present, and future. Loris 22: 38–44.
Fernando, A. & C.H. Fernando (1961). Report on the helminth
parasites of an Asian Elephant which died in
Singapore. Ceylon Veterinary Journal 9(4):
99–106.
Fernando, P., J. Jayewardene, T. Prasad, W. Hendavitharana & J. Pastorini
(2011). Current
status of Asian elephants in Sri Lanka. Gajah
35: 93–103.
Fleischer, R.C., E.A. Perry, K. Muralidharan,
E.E. Stevensand & C.M. Wemmer
(2001). Phylogeography of the Asian Elephant (Elephas maximus)
based on mitochondrial DNA. Evolution 55(9):
1882–1892.
Fontanarrosa, M.F., D. Vezzan,
J. Basabe & D.F. Eiras
(2006). An epidemiological
study of gastrointestinal parasites of dogs from Southern Greater Buenos Aires
(Argentina): age, gender, breed, mixed infections, and seasonal and spatial
patterns. Veterinary Parasitology 136: 283–295; http://doi.org/10.1016/j.vetpar.2005.11.012
Fowler, M.E. & S.K. Mikota (2008). Biology, Medicine, and Surgery
of Elephants. Blackwell Publishing Ltd, Oxford, UK.
Gaur, S.N.S., M.S. Sethi,
H.C. Tewari & O. Prakash
(1979). A
note on the prevalence of helminth parasites in wild
and zoo animals in Uttar Pradesh. Indian Journal of
Animal Science 49(2): 159–161.
Gupta, S.M.R. (1974). A preliminary report on diseases
and parasites of zoo animals, birds and reptiles. Indian
Journal of Animal Health 13(1): 15–24.
Heinrich, L. (2016). Prevalence and molecular
identification of helminths in wild and captive Sri
Lankan Elephants, Elephas maximus maximus. Future
19: 21.
Hing, S., N. Othman, S.K.
Nathan, M. Fox, M. Fisher & B. Goossens (2013). First parasitological survey of Endangered
Bornean elephants Elephas
maximus borneensis. Endangered Species
Research 21(3): 223–230.
Ihler, C.F., V. Rootwelt,
A. Heyeraas & N.I. Dolvik
(1995). The
prevalence and epidemiology of Anoplocephala perfoliata infection in Norway. Veterinary
Research Communications 19(6): 487–494; https://doi.org/10.1007/BF01839337
IUCN (2008). The IUCN
Red List of Threatened Species. http://doi.org/10.2305/IUCN.UK.2008.RLTS.T7140A12828813.en,
Electronic Version accessed on 10 September 2015.
IUCN (2017). The IUCN
Red List of Threatened Species. http://www.catalogueoflife.org/col/details/species/id/6fb47237753f0901b5bb779c6e9e1369/source/tree,
Electronic Version accessed on 29th January 2018.
Jayewardene, J. (1994). The Elephant in Sri Lanka.
Wildlife Heritage Trust of Sri Lanka, Colombo, 112pp.
Kuruwita, V.Y. & V.W.S.M. Vasanthathilake (1993). Gastro-intestinal [sic] nematode
parasites occurring in free-ranging Elephants (Elephas
maximus ceylonicus) in
Sri Lanka. Sri Lanka Veterinary Journal 40:
15–23.
Leimgruber, P., J.B. Gagnon, C. Wemmer,
D.S. Kelly, M.A. Songer & E.R. Selig (2003). Fragmentation of Asia’s remaining wildlands: implications for Asian Elephant conservation. Animal
Conservation 6: 347–359; http://doi.org/10.1017/S1367943003003421
Majewska, A.C., W. Kasprzak
& A. Werner (1997). Prevalence of Cryptosporidium in mammals housed in
Poznan Zoological Garden, Poland. Acta Parasitologica 42: 195–198.
Matsuo, K., S. Hayashi & M. Kamiya (1998). Parasitic infections of Sumatran Elephants in the Way Kambas National Park, Indonesia. Japanese Journal of
Zoo and Wildlife Medicine 3: 95–100.
Mbaya, A.W., G.K. Chuchan, F. Ballah & B. Garba (2012). Prevalence of Helminthic Infections among Wild Animals
in Yankari Game Reserve, Nigeria. Bulletin of Animal
Health and Production in Africa 60(1): 45–55.
Mbaya, A.W., M. Ogwiji & H.A. Kumshe (2013). Effects of host
demography, season and rainfall on the prevalence and parasitic load of
gastrointestinal parasites of free-living Elephants (Loxodonta
africana) of the Chad Basin National Park,
Nigeria. Pakistan Journal of Biological Sciences 16(20):
1152–1158.
Miller, D., B. Jackson, H.S. Riddle, C. Stremme, D. Schmitt & T. Miller (2015). Elephant (Elephas
maximus) health and management in Asia:
variations in veterinary perspectives. Veterinary Medicine International
2015(1): 2–3; http://doi.org/10.1155/2015/614690
Nilsson, H.O., P. Aleljung,
I. Nilsson, T. Tyszkiewicz & T. Wadström (1996). Immunomagnetic bead enrichment and PCR for detection of Helicobacter pylori
in human stools. Journal of Microbiological Methods
27(1): 73–79.
Nishanth, B., S.R. Srinivasan,
M.G. Jayathangaraj & R. Sridhar (2012). Incidence of endoparasitism in free-ranging elephants of Tamil Nadu
State. Tamilnadu
Journal of Veterinary & Animal Sciences 8(6): 332–335.
Nunn, C.L., S. Altizer, K.E.
Jones & W. Sechrest (2003). Comparative tests of
parasite species richness in primates. The American Naturalist
162(5): 597–614; http://doi.org/10.1086/378721
Obanda, V., T. Iwaki, N.M. Mutinda & F. Gakuya (2011). Gastrointestinal parasites and associated
pathological lesions in starving free-ranging African Elephants. South African
Journal of Wildlife 41: 167–172.
Perera, B.V.P. & R.P.V.J. Rajapakse (2009). Mortality and morbidity of wild
Elephants (Elephas maximus
maximus) of Sri Lanka, as a result of Liver
Flukes (Fasciola jacksoni)
infestation. Proceedings of the International Conference on Diseases of
Zoo and Wild Animals, Leibniz Institute for Zoo and Wildlife Research,
Beekse Bergen, The Netherlands.
Perera, K.U.E., S. Wickramasinghe, B.V.P. Perera,
& R.P.V.J Rajapakse (2014). Evaluation of morphometric features and
molecular characterization of its 2 and 28s genes of Anoplocephala
sp. from a Sri Lankan Elephant.
Proceedings of the Peradeniya University
International Research Sessions 18: 505; http://doi.org/10.4038/cjs.v45i1.7367
Rahman, S.M., A.R. Dey,
U.K. Kundu & N. Begum (2014). Investigation of
gastrointestinal parasites of herbivores at Dhaka National Zoological Garden of
Bangladesh. Journal of Bangladesh Agricultural
University 12(1): 79–85.
Samuel, W.M., A.A. Kocan
& M.J. Pybus (2001). Sarcoptes scabiei and sarcoptic
mange. Parasitic Diseases of Wild Mammals 2:
416–459.
Santiapillai, C. & R. Sukumar (2006). An overview of the status of the
Asian Elephant. Gajah 25: 3–8.
Santiapillai, C. & S. Wijeyamohan (2013). The First National Survey of Elephants in Sri Lanka. Current Science
105(2): 153–154.
Saseeendran, P.C., S. Rajendran,
H. Subramanian, M. Sasikumar, G. Vivek
& K.S. Anil (2003).
Incidence of helminthic infection among annually dewormed captive elephants.
Zoos Print Journal 19(3): 1422; http://doi.org/10.11609/JoTT.ZPJ.19.3.1422
Scott, M.E. & A. Dobson (1989). The role of parasites
in regulating host abundance. Parasitology Today
5: 176–183.
Seneviratna, P. (1955). A checklist of helminths
in the Department of Veterinary Pathology, University of Ceylon, Peradeniya. Ceylon Veterinary
Journal 3: 32–37.
Seneviratna, P. & J.B. Jayasinghe (1968). Some parasites from the Ceylon
Elephants (Elephas maximus).
Ceylon Veterinary Journal 15:
28.
Soulsby, E.J.L. (1982). Helminths, Arthropods and Protozoa of Domesticated Animals. Bailliere Tindall, London, 809pp.
Suresh, K., P.C. Choudhuri,
K.A. Ajithkumar, M.D. Hafeez
& P.A. Hamza (2001). Epidimiological and clinico
- therapeutic studies of strongylosis in elephants. Zoos Print Journal 16(7):
539–540; http://doi.org/10.11609/JoTT.ZPJ.16.7.539-40
Tiwari, S.P. & K.N.P. Rao (1996). Fenbendazole - a useful dewormer for elephants. Zoo’s Print 9(4):
2–5.
Vanitha, V., K. Thiyagesan & N. Baskaran
(2011). Social life of
captive Asian Elephants (Elephas maximus) in Southern India: implications for elephant
welfare. Journal of Applied Animal Welfare Science
14(1): 42–58.
Varadharajan, A. & A. Kandasamy (2000). A survey of gastro-intestinal
parasites of wild animals in captivity in the V.O.C. Park and Mini Zoo,
Coimbatore. Zoos’ Print Journal 15(5): 257–258; http://doi.org/10.11609/JoTT.ZPJ.15.5.257-8
Vidya, T.N.C. & R. Sukumar (2002). The effect of some ecological factors on the
intestinal parasite loads of the Asian Elephant (Elephas
maximus) in southern India. Journal of
Biosciences 27(5): 521–528; http://doi.org/10.1007/BF02705050
Warren, K., J. Bolton, R. Swan, W. Gaynor & L.
Pond (1996). Treatment
of gastrointestinal tract impaction of a 2-year-old Asian Elephant (Elephas maximus).
Australian Veterinary Journal 73: 37–38; http://doi.org/10.1111/j.1751-0813.1996.tb09956.x
Watve, M.G. (1995). Helminth parasites of elephants. Ecological
aspects, pp. 289–295. In: Daniel, J.C. & H. Datye (eds.). A Week with Elephants
Bombay. Oxford University Press, Bombay Natural History Society, New
Delhi.
World Health Organization (WHO) (1991). Basic laboratory methods in
medical parasitology. Geneva: World Health Organization. Downloaded
14 March 2015.