Journal of
Threatened Taxa | www.threatenedtaxa.org | 26 September 2018 | 10(10):
12337–12343
Comparison of beach profiles conducive for turtle nesting in
Andaman
Subramanian Narayani 1, Sasidharan Venu 2 & Andrea Joan D’Silva
3
1,2,3 Department of Ocean Studies and
Marine Biology, Pondicherry University, Post Bag No. 1, Brookshabad
Campus, Chakkargaon Post, Port Blair, Andaman &
Nicobar 744112, India
1 nans.mythila@gmail.com (corresponding author), 2 s.venu1974@gmail.com,
3 andreajdsilva@gmail.com
doi: https://doi.org/10.11609/jott.3373.10.10.12337-12343
Editor: B.C. Choudhury, Wildlife
Trust of India, Noida, India. Date of
publication: 26 September 2018 (online & print)
Manuscript details: Ms # 3765 |
Received 25 February 2017 | Final received 30 August 2018 | Finally accepted 15
September 2018
Citation: Narayani, S., S. Venu
& A.J. D’Silva (2018).
Comparison of beach
profiles conducive for turtle nesting in Andaman. Journal of Threatened
Taxa 10(10): 12337–12343; https://doi.org/10.11609/jott.3373.10.10.12337-12343
Copyright: © Narayani et al. 2018. Creative Commons Attribution 4.0
International License. JoTT allows
unrestricted use of this article in any medium, reproduction and distribution
by providing adequate credit to the authors and the source of publication.
Funding: None.
Competing interests: The authors declare no competing interests.
Author Details: Dr. S. Narayani has completed her PhD from Pondicherry University on the feeding ecomorphology of reef fishes from Andaman. She is
interested in fisheries ecology, conservation biology and behavioural
ecology. Dr. S. Venu is currently working
as Assistant Professor in the Department of Ocean Studies and Marine Biology,
School of Life Sciences, Pondicherry University at Port Blair Campus. Present
research interests include fish taxonomy & molecular phylogeny, fishery
biology & ecomorphology, coral reef resilience,
fishing technology & landings. Ms.
Andrea Joan D’Silva has completed MSc in
Marine Biology from Pondicherry University in Andaman Campus. She is interested
in conservation biology and conservation education. She is now working in the
education sector.
Author
Contributions: SN assisted in field
surveys and prepared the manuscript. SV conceived and designed the work and finalised the drafts. AJD’S carried out the field surveys
and assisted in the manuscript writing.
Abstract: The present study was undertaken to
compare beach characteristics associated with turtle nesting in the Andaman
group of islands. Karmatang,
Kalipur, Ramnagar, Chidiyatapu, Carbyn’s Cove, and Wandoor were chosen as study sites. Beach slope, sand grain
characteristics, and general vegetation patterns were analysed. The angle of inclination of the beach slope
ranged from 2.06 to 8.3 degrees. Beaches
with a higher angle had a comparatively higher number of nesting sites. The study shows that a single factor does not
make a beach more conducive for nesting.
Chidiyatapu has the widest beach but lacks
other features and so it is not a preferred nesting site. The grain size of sand in Wandoor
is highly favourable, but the intertidal region is
not long and there are streams that can drown the nests. Karmatang has a
long beach and a higher slope angle. Ramnagar has a moderate beach length and a high slope
angle. The dominant grains at both the
beaches were found to be granules. The
absence of streams and artificial light, fewer number of anthropogenic
activities, lack of obstacles, the presence of bordering vegetation, and a
conducive beach slope with granular sand grains make Ramnagar,
Karmatang, and Kalipur
ideal for turtle nesting.
Keywords: Sand grain, beach slope,
intertidal, ecology, beach angle, turtle nesting, Andaman.
Introduction
Among
the many species that appeared as part of the modern marine turtle families in
the Cretaceous (Lutz & Musick 1996), only seven
species remain today. Among these, one
is endangered, three are vulnerable, two are critically endangered and one is
listed as data deficient (Nicholas 2001; IUCN 2018). Five species are reported from India and four
species are reported from Andaman & Nicobar Islands (Murugan 2010).
Selection of a good nesting site is an important stage for oviparous
animals, especially in those species that do not provide parental care
(Morales-Mavil et al. 2016). Minimizing female mortality and maximizing
offspring fitness are the driving forces for site selection by female turtles
for nesting (Spencer 2002).
The Andaman & Nicobar
archipelago is located in the Bay of Bengal between 6.750o–13.750o N
& 92.000o–94.300o E, extends over 800km, and consists
of islands, islets, and rocky outcrops with a coastline stretch of 1962km. Four species of marine turtles occur in the
Andaman & Nicobar Islands: Leatherback Dermochelys
coriacea, Hawksbill Eretmochelys
imbricata, Green Turtle Chelonia
mydas, and Olive Ridley Lepidochelys
olivacea.
These turtles are protected under Schedule I of the Indian Wildlife
(Protection) Act, 1972. The ban on
hunting and harvesting of turtles was enforced in 1977, but the indigenous
groups of the Andaman & Nicobar Islands are exempt from the Act as marine
turtles have been their source of food for centuries (Bhaskar
1984). The surveys and studies conducted
in the Andaman & Nicobar Islands have recorded India’s best nesting beaches
for Leatherback, Hawksbill, and Green turtles (Andrews et al. 2006). The present study was undertaken to review
the status of marine turtles in Andaman and to compare the beach characteristics
associated with turtle nesting.
Materials And Methods
The study was conducted during
February–March 2014.
Study area
Six stations in the Andaman Islands
were selected as study sites for this work.
Karmatang Beach at 12.9130N &
92.8960E is a bay located in Mayabunder,
North Andaman (Fig. 1). It is a sandy
beach that is dark-coloured, giving the water a very
turbid look. Good vegetation, with a mix
of shrubs and trees, lines the beach. Ramnagar is situated in Diglipur,
North Andaman, and is located at 13.0750N & 93.0280E. This sandy beach is 15km away from Kalighat. It is surrounded by palm and coconut trees and coastal shrubs. Comparatively, it has stronger waves than the
other study stations. Kalipur is located in Diglipur,
North Andaman, and it is the only beach in the world where four species of
turtles come to nest. Its coordinates
are 13.2350N & 93.8960E and it is 18km from Diglipur. It has a
combination of sand and rocks. Chidiyatapu houses the Munda Pahar Beach, which is 2.5km from Chidiyatapu
Beach. Its geographical coordinates are
11.4900N & 92.7080E.
The beach has a combination of sand and rocks and has small freshwater
sources. Carbyn’s
Cove is a bay that is on the southeast of South Andaman. It is located at 11.4900N &
92.7000E. It is a sandy beach
with rocks flanking its sides. There is
an estuary adjoining it that supports a healthy
mangrove vegetation. Wandoor
is a marine national park located 29km from the city of Port Blair and is
situated in the Bay of Bengal. It is
located at 11.6090N & 92.6750E. It is a white sandy beach with two small
freshwater inlets. It has a good
surrounding vegetation of shrubs, mangroves, and woody trees.
The slope of the beach
The slope of the beach was estimated
by employing the method described by Varela-Acevedo et al. (2009) using Auto
Level, DSZ2 (manufactured by Suzhou FOIF Co. Ltd.). The distance between the scale and the
telescope was calculated. The values of
distance against height were plotted on a graph to obtain the beach profile.
The angle of inclination
By finding the slope of the land,
the height of the land was found at certain distances. Using the values of height and distance in
the trigonometric formula tan ѳ, the value for the angle of inclination was obtained.
Grain size analysis
The grain size of the sand on the
beach was analysed following Varela-Acevedo et al.
(2009). Using a corer of length 12.7cm
and a width of 5.08cm, sand samples were obtained from the part of the beach
that is higher than the tide mark. None of these parts were in the dune area as
there are no dunes in Andaman. The
collected samples were placed in sample bags for analysis. The grains were mixed well and sprinkled onto
a slide with a layer of oil to adhere to the grains. The grains were then viewed under a polarising microscope that was fixed with a graduated
ocular lens. The diameter of each grain
was measured individually in divisions and converted to millimetres. In each sand sample, diameters of 170 grains
were measured. Size class intervals and
their corresponding frequencies were made and the results were depicted
graphically. The class interval with the
highest frequency was taken as the representative of the sand at that
corresponding sampling site. The sand
grains were classified based on Wentworth (1922).
Extrinsic parameters
By comparing the vegetation at each
of the six sites, the amount of vegetation at each site was classified as high,
medium, or low. The presence of
obstacles like trees was noted by visual examination. Anthropogenic activities/ influences like
manmade structures, vehicles, shacks, and pollution were taken into account
through comparison among the study stations.
Techniques for identifying key parameters and estimating their values
were followed from Varela-Acevedo et al. (2009). The transformed data of extrinsic parameters
and the presence of turtle nests reported from literature (Andrews 2006; Murugan 2010) were used to perform principal component
analysis and to generate a plot in PRIMER E-V6 package (Clark & Warwick
2001).
Results
Extrinsic parameters
The extrinsic parameters are given in Table 1.
Beach slopes
Karmatang has a relatively flat reef slope (Fig. 2) with a minor dip at
2.4m and a major dip at 13.5m. The
profile of Kalipur (Fig. 3) is very undulating with
only one major visible rise at 31.4m. Ramnagar has a major rise at 7m and another at 12m (Fig.
4). The profile of Chidiyatapu
(Fig. 5) shows that it has a number of indentations that can be difficult for
turtles to navigate. From the profile of
Carbyn’s Cove (Fig. 6), it can be seen that there is only one major dip at 7m but otherwise, the land is relatively flat.
In the case of Wandoor Beach (Fig. 7), there
is a rise at 5.5m and a minor dip at 11.5m, but otherwise, the land is without
many undulations. Chidiyatapu
is the widest beach while Wandoor is the narrowest
(Fig. 8).
The angle of the slope
The slope angles of the study
stations are presented in Table 1.
Sand grain analysis
In Karmatang,
the majority of sand grains were small in size.
This was the case in Kalipur and Ramnagar as well. In
Chidiyatapu, the majority of sand grains were in the
middle-size category. In Carbyn’s Cove and Wandoor, the
sand grains were small in general.
According to the classification of sand grains by Wentworth (1922), Karmatang and Kalipur have very
coarse grains, Chidiyatapu and Wandoor
have granules, and Carbyn’s Cove has pebbles (Table
2). Overall, the majority of grains were
in the size range 2–4 mm. Wandoor and Karmatang had a more
or less equitable distribution of sand grain sizes. There were proportionally more
larger grains in Carbyn’s cove and more
smaller grains in Kalipur.
Effect of extrinsic parameters
With all the parameters mentioned
above, Table 3 (++ very favourable, +favourable, - not favourable)
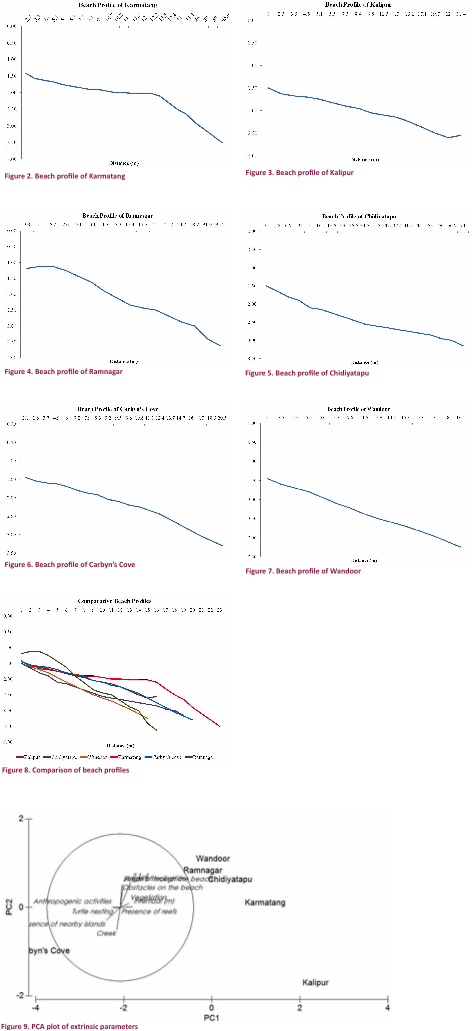
provides a comparison of the study areas to show the effect of the analysed parameters on turtle nesting. The principal component analysis (Fig. 9)
revealed that the absence of anthropogenic activities and nearby islands and
the absence of creeks were closely associated with turtle nesting in the
stations. The first two principal
components accounted for 82% of the total variation. It is acknowledged here that if the specific
number of nests in each area is included in the analysis, these results may
vary. This is especially true of regions
like Chidiyatapu and Wandoor
for which results are only available from pre-Tsunami surveys.
Discussion
Sea turtle populations have
decreased due to habitat destruction, anthropogenic activities on nesting
beaches, predation of young hatchlings, and theft of unhatched
eggs (Wyneken et al. 1988). The major potential terrestrial factors for
choosing a beach for nesting are beach slope and width, the presence of
interspecific competition, artificial lighting, and human activities. Studies have shown that there is a positive
feedback between turtles and the beach dunes in which they nest (Bouchard &
Bjorndal 2000).
Beaches with good access to the sea, fine sands of small grain size, and
adequate humidity and temperature were previously noted as the desired features
for site selection for turtle nesting (Wood & Bjorndal
2000; Morales-Mavil 2016).
The location of the nest in the
tidal zone is crucial as the eggs must neither be flooded and eroded nor be
exposed to land predators (Whitmore & Dutton 1985; Blamires
& Guinea 1998). Hatchlings must be
able to find the sea and the nest must not have visual obstructions that
prevent the same (Godfrey & Barreto 1995). This shows that Wandoor,
with the smallest beach width among all study stations, is not favourable for turtle nesting.
Debris on the beach prevents
successful nesting and causes a phenomenon called as ‘false crawl’ where the
females emerge from the water but do not deposit an egg clutch (Fujisaki &
Lamont 2016). Artificial lighting too
has been reported to disrupt patterns of nesting females (Weishampel
et al. 2016).
Large angled beaches are preferred
by turtles as water cannot move up the slope as easily and hence the nests are
relatively safer from flooding (Godley et al. 1993). Ramnagar and Karmatang beaches have the steepest profile and larger
angles, and so they are very favourable for turtle
nesting. Ramnagar
has the highest dominance of granules, which seem to be the ideal grain size as
supported by the results from Hughes et al. (2009) that show that real nest
contains medium sand or larger grains.
Though Chidiyatapu has the widest beach, other
factors are not very favourable and this leads to
only sporadic nesting. Wandoor has the required grain size but the lack of
intertidal width and the presence of streams in the beach are deterring
factors. Considering all the features
studied, the absence of streams, absence of artificial light, a significantly
lesser number of anthropogenic activities, lack of obstacles, and the presence
of bordering vegetation make Karmatang, Kalipur, and Ramnagar very
conducive for turtle nesting. It has
been reported that a total of 99 nesting sites belonging to four species of
turtles were seen in Ramnagar, Karmatang,
and Carbyn’s Cove (Andrews 2006). While it could be deduced from the present
study that Kalighat is a beach conducive for turtle
nesting, the evidence for turtle nesting in this beach is mainly
anecdotal. Unfortunately, data from the
literature for these stations is sporadic.
It is acknowledged here that a comprehensive list of sea turtle nests in
these stations could be useful in comparing predicted conduciveness and actual
preference. It is hereby recommended
that the number of nests along each beach in these stations is
to be quantified to empirically ascertain nesting preferences of turtles in
this region.
The spatial and temporal consistency
of turtle nesting behaviour are of basic importance
to conservation efforts as they can be used to interpret scales of behavioural patterns in relation to environmental
parameters. This can be used to regulate
human activities in the beaches where turtles nest regularly (Weishampel et al. 2016).
There are numerous studies all
around the world regarding turtle nesting site selection, environmental
criteria for embryonic development, and other aspects of sea turtle
biology. The focal point of all these
studies is that a better understanding of the biology and life history of
turtles can help in planning more effective conservation strategies. When compared to other regions, the studies
regarding turtles from Andaman & Nicobar are meagre. Further research can point out the salient
features of turtle nesting behaviour in these regions
and they can be used for the conservation of these marine reptiles.
Conclusion
Turtles have been part of Andaman’s
history since the 1800s. Their constant
association with these waters and their homing in annually provides the best
evidence that the beaches in Andaman do meet the turtles’ requirements. This study shows that a single factor does
not make a beach a better nesting site.
It is shown from this study that there is a significant lack of
literature pertaining to the reproductive biology of turtles in these islands. The results from further studies can be a
backbone for planning developmental activities and developing infrastructure
for these beaches in the future.
Table 1. Extrinsic parameters in the
study sites
|
Parameters |
Karmatang |
Kalipur |
Ramnagar |
Chidiyatapu |
Carbyn’s Cove |
Wandoor |
|
Intertidal (m) |
53.6 |
32.9 |
21.3 |
73.5 |
15.6 |
14.9 |
|
Vegetation |
High |
High |
High |
High |
Low |
Moderate |
|
Streams through the beach |
Nil |
Nil |
Nil |
2 |
Nil |
2 |
|
Creek |
Nil |
1 |
Nil |
Nil |
1 |
Nil |
|
Obstacles on the beach |
Nil |
Nil |
Nil |
Yes |
Nil |
Yes |
|
Presence of nearby islands |
Nil |
Nil |
Nil |
Nil |
2 |
Nil |
|
Presence of reefs |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
|
Anthropogenic activities |
Low |
Low |
Low |
Moderate |
High |
Moderate |
|
Angle of inclination |
7.86o |
2.062o |
8.3o |
2.75o |
2.29o |
5.71o |
Table 2. Grouping of grains based on
size classification by Wentworth (1922)
|
|
Karmatang |
Kalipur |
Ramnagar |
Chidiyatapu |
Carbyn |
Wandoor |
Classification |
|
0–1 |
0 |
1 |
0 |
0 |
0 |
0 |
Coarse sand |
|
1.0–2.0 |
76 |
112 |
17 |
12 |
2 |
36 |
Very coarse sand |
|
2.1–4 |
75 |
55 |
106 |
142 |
28 |
78 |
Granules |
|
4.1–16 |
19 |
2 |
47 |
16 |
124 |
56 |
Pebbles |
|
>16.1 |
0 |
0 |
0 |
0 |
16 |
0 |
Gravel |
Table 3. Effect of extrinsic parameters
(++ very favourable, +favourable, - not favourable)
|
Site |
Karmatang |
Kalipur |
Ramnagar |
Chidiyatapu |
Carbyn’s Cove |
Wandoor |
|
Sand grains |
+ |
++ |
++ |
- |
- |
++ |
|
Beach width |
+ |
+ |
+ |
++ |
- |
- |
|
Stream/ creek |
+ |
+ |
+ |
- |
+ |
- |
|
Presence of obstacles |
+ |
+ |
+ |
- |
- |
- |
|
Artificial light |
+ |
+ |
+ |
+ |
- |
+ |
|
Vegetation |
++ |
++ |
++ |
++ |
- |
+ |
|
Anthropogenic activity |
++ |
++ |
++ |
+ |
- |
+ |
References
Andrews, H.V., A. Tripathy, S. Aghue, S. Glen, J.
Saw & K. Naveen (2006). The status
of sea turtle populations in the Andaman and Nicobar Islands of India.
In: K. Shanker & H.V. Andrews (eds). Towards an Integrated and Collaborative Sea Turtle
Conservation in India: a UNEP/CMS-IOSEA
Project of Priority Research Areas. Center for Herpetology/Madras
Crocodile Bank Trust., Chennai. 92 pp.
Bhaskar, S. (1984). Distribution
and status of sea turtles in India, pp21–35. In: Silas, E.G. (ed.). Proceedings of the Workshop on Sea Turtle Conservation.
CMFRI, Cochin, Special Publication No. 18, 120pp.
Bhasker, S. (1979). Sea
turtle survey in the Andamans and Nicobars.
Hamadryad 4(3): 2–26.
Blamires, S.J. & M.L. Guinea (1998). Implications of nest site selection on egg predation at the sea
turtle rookery at Fog Bay, pp22–24. In: Kennett R., A. Webb, G. Duff, M.L. Guinea & G.J.E. Hill (eds.). Proceedings
of the Marine Turtle Conservation and Management in Northern Australia Workshop.
Centre for Indigenous and Natural Resources, Centre for Tropical Wetlands
Management, Darwin, 89 pp.
Bouchard, S.S. & K.A. Bjorndal
(2000). Sea turtles as biological
transporters of nutrients and energy from marine to terrestrial ecosystems. Ecology 81: 2305–2313.
Clarke, K.R. &
R.M. Warwick (2001). Change in marine communities: an approach to
statistical analysis and interpretation, 2nd edition. PRIMER-E,
Plymouth, 172pp.
Dattatri, S. (1984). Threats
to sea turtles in India- exploitation and habitat perturbations, pp.59–66.
In: Silas, E.G. (ed.). Proceedings of the Workshop on Sea Turtle Conservation.CMFRI,
Cochin, Special Publication No. 18, 120pp.
Fujisaki, I. & M.M. Lamont (2016). The effects of large beach debris on nesting
sea turtles. Journal of Experimental Marine Biology
and Ecology 482: 33–37.
Godfrey, M.H. &
R. Barreto (1995). Beach vegetation and sea-finding orientation of turtle hatchlings.
Biological Conservation 74: 29–32.
Godley, B.J., A.C.
Broderick, S. Blackwood, L. Collins, K. Glover, C. McAldowie,
D. McCulloch & J. McLeod (1993). 1991
survey of marine turtles nesting in Trinidad and Tobago. Marine Turtle Newsletter 61: 15–18.
Hughes, G.N., W.F.
Greaves & J.D. Litzgus (2009). Nest selection by Wood Turtles (Glyptemys insculpta)
in a thermally limited environment. Northeastern Naturalist 16(3):
321-338.
IUCN (2018). The IUCN Red list of Threatened Species. Version 2018-1. http://www.iucnredlist.org
Electronic version accessed on 18.9.2017.
Kar, C.S. & M.C. Dash (1984). Mass nesting beaches of the olive ridley Lepidochelys olivacea (Eschscholtz, 1829)
in Orissa and behavior during an arribada,
pp.36–48. In: Silas, E.G. (ed.). Proceedings of the
Workshop on Sea Turtle Conservation. CMFRI,
Cochin, Special Publication No. 18, 120pp.
Lutz, P.L. &
J.A. Musick (eds.) (1996). The Biology of Sea Turtles. CRC Press, USA, 448pp.
Mohan, L.R.S.
(1986). Observations on the ecology
of the nest and on some aspects of reproductive behaviour
of the Ridley Turtle Lepidochelys olivacea from Calicut Coast. Indian
Journal of Fisheries 33(1): 39–44.
Morales-Mavil, J.E., L.A.
Contreras-Vega, A. Serrano, J. Cobos-Silva & L. Zavaleta-Lizárraga (2016). Spatial-temporal distribution of Kemp’s Ridley Turtles (Lepidochelys kempi)
and Green Turtles (Chelonia mydas) nests in a beach of the north of Veracruz,
Mexico, pp.33–53. In: Patterson, C. (ed.) Sea Turtles: Ecology, Behavior and
Conservation. Nova Publishers, New York, 147 pp.
Murugan, A. (2010). The past and present scenario of sea turtles in India: an
overview of possibility for recurrence of history. Proceedings of the 5th
International Symposium on SEASTAR2000 and Asian Bio-logging Science (The 9th
SEASTAR2000 workshop): 33–35;
https://repository.kulib.kyoto-u.ac.jp/dspace/bitstream/2433/107339/1/9thSeastar_33.pdf
Nicholas, M.
(2001). Light pollution and marine turtle hatchlings:
the straw that breaks the camel’s back? George Wright
Forum 18(4): 77–82.
Spencer, R-J.
(2002). Experimentally testing nest site selection:
fitness trade-offs and predation risk in turtles. Ecology 83(8):
2136–2144; https://doi.org/10.1890/0012-9658(2002)083[2136:ETNSSF]2.0.CO;2
Switzer, P.V.
(1993). Site fidelity in predictable
and unpredictable habitats. Evolutionary Ecology 7(6): 533–555.
Varela-Acevedo, E., K.L. Eckert, S.A. Eckert, G. Cambers &
J.A. Horrocks (2009). Sea turtle nesting beach characterization
manual, pp.46–97. In: Examining the Effects of Changing Coastline
Processes on Hawksbilll Sea Turtle (Eretmochelys imbricata) Nesting
Habitat. Master’s Project, Nicholas School of the
Environment and Earth Sciences, Duke University. Beaufort,
North Carolina USA, 97pp.
Weishampel, J.F., D.A.
Bagley, L.M. Ehrhart & B.L. Rodenbeck
(2003). Spatiotemporal patterns of
annual sea turtle nesting behaviors along an East Central Florida beach.
Biological Conservation 110: 295–303.
Weishampel, Z.A., W-H. Cheng & J.F. Weishampel (2016). Sea turtle nesting patterns in Florida
vis-à-vis satellite-derived measures of artificial lighting. Remote
Sensing in Ecology and Conservation 2(1): 59–72; http://doi.org/10.1002/rse2.12
Wentworth, C.K.
(1922). A scale of grade and class
terms for clastic sediments. The Journal of Geology 30(5): 377–392.
Whitmore, C.P. & P.H. Dutton (1985). Infertility, embryonic mortality and nest-site selection in
leatherback and green sea turtles in Suriname. Biological Conservation 34(3): 251–272.
Wood, D.W. & K.A. Bjorndal (2000). Relation of temperature, moisture, salinity,
and slope to nest site selection in Loggerhead Sea Turtles. Copeia
2000(1): 119–128.
Wyneken, J., T.J. Burke, M. Salmon & D.K. Pederson (1988). Egg failure in natural and relocated sea turtle nests. Journal of Herpetology 22(1): 88–96.